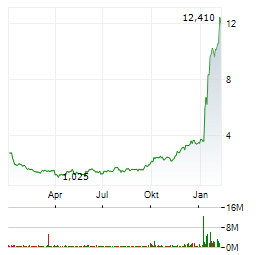
| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
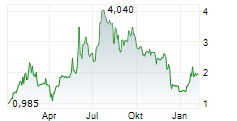
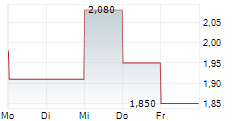
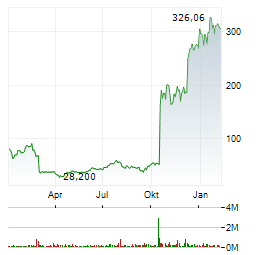
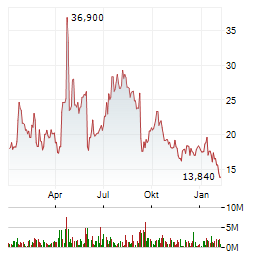
| 1,640 | 1,780 | 02.06. |
Chart-Typ:
Zeitraum:
| Praxis Precision Medicines, Inc.: Praxis Precision Medicines Provides Vormatrigine Program Update | POWER1 Study in highly refractory patients with focal onset seizures did not meet its primary success measure Secondary measure, the 50% response rate, was met and seizure reduction during the second... ► Artikel lesen |
| Praxis Precision Medicines, Inc.: Praxis Precision Medicines Provides Corporate Update and Reports First Quarter 2026 Financial Results | FDA accepted the new drug application (NDA) for ulixacaltamide in Essential Tremor with a PDUFA target action date of January 29, 2027, and the NDA for relutrigine, with priority review, in SCN2A... ► Artikel lesen |
| Praxis Precision Medicines, Inc.: Praxis Precision Medicines Announces FDA Acceptance of New Drug Application for Ulixacaltamide HCl in Patients with Essential Tremor | FDA assigned PDUFA target action date of January 29, 2027 No advisory committee meeting expected BOSTON, April 14, 2026 (GLOBE NEWSWIRE) -- Praxis Precision Medicines, Inc. (NASDAQ: PRAX), a fully... ► Artikel lesen |
| Erasca, Inc.: Erasca Reports First Quarter 2026 Business Updates and Financial Results | Robust monotherapy efficacy and generally well-tolerated safety results observed during dose escalation for ERAS-0015 in both KRAS G12X NSCLC and PDAC reinforce best-in-class potential across RAS-targeted... ► Artikel lesen |
| Erasca, Inc.: Erasca Announces Clinical Trial Collaboration and Supply Agreement with Merck to Evaluate ERAS-0015 in Combination with KEYTRUDA (Pembrolizumab) | SAN DIEGO, May 11, 2026 (GLOBE NEWSWIRE) -- Erasca, Inc. (Nasdaq: ERAS), a clinical-stage precision oncology company singularly focused on discovering, developing, and commercializing therapies for... ► Artikel lesen |
| Erasca, Inc.: Erasca Announces Positive Preliminary Phase 1 Dose Escalation Data for Potentially Best-in-Class Pan-RAS Molecular Glue ERAS-0015 in KRAS-Mutant Solid Tumors | Robust monotherapy efficacy in KRAS G12X NSCLC: 62% uORR in 2L+ and 75% uORR in post-ICI/platinum 2/3L at 16-32 mg QD PAD, and 64% uORR in 2L+ at 24-32 mg QD RDE Robust monotherapy efficacy in 2L... ► Artikel lesen |
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu HEALIOS KK finden Sie auf Wallstreet Online.