SARM1 knock-out in an EAE mouse model demonstrates evidence of axonal protection
Disarm Therapeutics, a biotechnology company creating a new class of disease-modifying therapeutics for patients with axonal degeneration, a central driver of neurological disease, today announced preclinical data demonstrating that deleting SARM1 has a robust axonal-protective effect in experimental autoimmune encephalomyelitis (EAE), a well-accepted animal model of multiple sclerosis. These data suggest that targeting SARM1 presents a novel, disease-modifying approach to treating multiple sclerosis. The findings are being presented as a late-breaking poster at the European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS) in Stockholm on Sept. 13, 2019.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20190911005148/en/
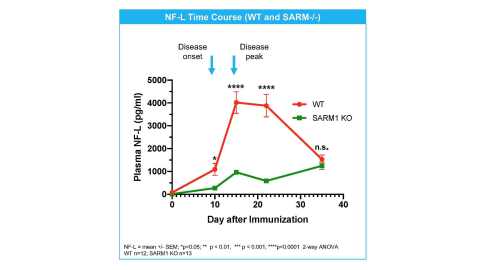
SARM1 genetic deletion reduces NfL and protects axons in EAE (Graphic: Business Wire)
"Today's results are particularly exciting because they provide further evidence that SARM1 is a viable therapeutic target for MS and other CNS axonopathies," said Dr. Alvin Shih, President and CEO of Disarm Therapeutics. "To date, treatments for patients with MS have focused on immunomodulatory therapies to slow disease. With these new data, there is potential for a new and complementary approach to modifying the course of disease in MS."
Disarm's findings include:
- In an EAE model, SARM1 homozygous and heterozygous gene deletion markedly attenuates the increase in plasma neurofilament light (NfL), a biomarker of axonal degeneration and disease progression in MS. The magnitude of SARM1 effect on NfL was gene-dosage dependent.
- Axonal protection through SARM1 deletion led to a statistically significant reduction in demyelination via histological examination.
- Disease onset, peak disease scores, and time course were not affected by SARM1 deletion, suggesting that the benefits of SARM1 genetic deletion are not due to interference with the inflammation-driven disease process in this EAE animal model of MS.
"These experimental results deepen our growing understanding of SARM1," added Rajesh Devraj, Ph.D., Chief Scientific Officer and co-founder of Disarm Therapeutics. "Previous research demonstrated that SARM1 inhibition could protect axons in traumatic injuries. These new data go further, establishing that SARM1 plays a role in protecting axons from immuno-inflammatory damage in diseases such as MS."
The poster, P1651, is part of Poster Session 3 on Sept. 13 from 12:15 to 2:15 p.m. CEST.
About Disarm Therapeutics
Disarm Therapeutics is a biotechnology company that is creating a new class of disease-modifying therapeutics for patients with axonal degeneration, a central driver of neurological disease-causing disability and disease progression. By inhibiting the SARM1 protein, identified by the company's scientific founders as the central driver of axonal degeneration, these therapeutics may prevent the loss of axons in chronic and acute diseases of the central, ocular, and peripheral nervous systems. For a broad range of diseases including multiple sclerosis, amyotrophic lateral sclerosis, glaucoma, and peripheral neuropathies, the therapeutic goal is to prevent further degeneration, stabilize disease, and allow for functional recovery. Disarm was founded by Atlas Venture, Dr. Jeffrey Milbrandt and Dr. Aaron DiAntonio of Washington University in St. Louis, and a team of exceptional scientists and drug developers committed to developing a new treatment paradigm for patients with neurological diseases. For more information, please visit www.disarmtx.com.
View source version on businesswire.com: https://www.businesswire.com/news/home/20190911005148/en/
Contacts:
Sara Green, Ten Bridge Communications
sgreen@tenbridgecommunications.com
617-233-1714
