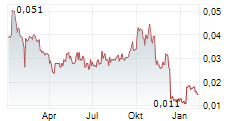
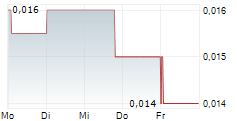
Highlights (unaudited):
- Consistent Revenue Growth: Immuron is no longer just a "concept" company. Global H1 FY26 sales grew to AUD$4.2 million, with the U.S. market showing strong momentum (+17% YoY).
- Cash (AUD $10.0 million) to fund operating activities. Includes AUD $7.3 million (net) raised at an average price of A$0.0803 (IMRN USD$2.0923).
- Validated Platform: The "Hyper-Immune" platform is versatile. Beyond traveler's diarrhea (Travelan), the company is moving into high-value clinical targets like C. diff (IMM-529)
- Partnerships: Immuron has taken a strategic decision to pursue partnerships to fund progress of Travelan(IMM-124E) and IMM-529 clinical programs.
- IMM-124E: Eligible for end of Phase 2 meeting with the U.S. Food and Drug Administration (FDA)
- IMM-529: Investigational New Drug (IND) Application approved by the FDA
MELBOURNE, Australia, Feb. 25, 2026 (GLOBE NEWSWIRE) -- Immuron Limited (ASX: IMC; NASDAQ: IMRN), an Australian based and globally integrated biopharmaceutical company today released half-year financial results and separately a presentation outlining its Strategic Reset.
A copy of the presentation slide deck is available on the Company's website:
https://investors.immuron.com.au/announcements/7397157
About Immuron
Immuron Limited (ASX: IMC, NASDAQ: IMRN), is an Australian biopharmaceutical company focused on developing and commercializing orally delivered targeted polyclonal antibodies for the treatment of infectious diseases.
About Travelan
Travelan is an orally administered passive immunotherapy that prophylactically reduces the likelihood of contracting travelers' diarrhea, a digestive tract disorder that is commonly caused by pathogenic bacteria and the toxins they produce. Travelan is a highly purified tabletized preparation of hyper immune bovine antibodies and other factors, which when taken with meals bind to diarrhea-causing bacteria and prevent colonization and the pathology associated with travelers' diarrhea. In Australia, Travelan is a listed medicine on the Australian Register for Therapeutic Goods (AUST L 106709) and is indicated to reduce the risk of Travelers' Diarrhea, reduce the risk of minor gastro-intestinal disorders and is antimicrobial. In Canada, Travelan is a licensed natural health product (NPN 80046016) and is indicated to reduce the risk of Travelers' Diarrhea. In the U.S., Travelan is sold as a dietary supplement for digestive tract protection.
Travelers' diarrhea (TD)
TD is generally defined as the passage of = 3 unformed stools per 24 hours plus at least one additional symptom (such as nausea, vomiting, abdominal cramps, fever, blood/mucus in the stools, or fecal urgency) that develop while abroad or within 10 days of returning from any resource-limited destinations (Leung et al., 2006). Diarrhea continues to be the most frequent health problem among travelers to destinations in lower- and middle-income regions (Steffen, 2017). Deployed US military personnel, essentially representing a long-term traveller population, are particularly affected given their population dynamics and the context in which they seek care and treatment (Connor et al., 2012). Diarrhea is the leading infectious disease threat to the overall health and preparedness of deployed US armed forces, with diarrheagenic E. coli, Campylobacter spp., and Shigella spp. among the most commonly reported etiologies (Riddle et al., 2006).
Immuron Platform Technology
Immuron's proprietary technology is based on polyclonal immunoglobulins (IgG) derived from engineered hyper-immune bovine colostrum. Immuron has the capability of producing highly specific immunoglobulins to any enteric pathogen and our products are orally active. Bovine IgG can withstand the acidic environment of the stomach and is resistant to proteolysis by the digestive enzymes found in the Gastrointestinal (GI) tract. Bovine IgG also possesses this unique ability to remain active in the human GI tract delivering its full benefits directly to the bacteria found there. The underlying nature of Immuron's platform technology enables the development of medicines across a large range of infectious diseases. The platform can be used to block viruses or bacteria at mucosal surfaces such as the Gastrointestinal tract and neutralize the toxins they produce.
IMM-124E (Travelan-
IMM-124E was developed using Immuron's platform technology. IMM-124E is produced from the colostrum of birthing cattle that have been immunised during pregnancy with a vaccine containing the outer antigens of multiple human derived ETEC. A total of 13 ETEC strains are used in the vaccine to produce high levels of antibodies against selected surface antigens from the most common strains of ETEC.
The resultant hyperimmune colostrum IMM-124E from ETEC vaccinated cows contains significant levels of polyclonal antibodies specific for ETEC antigens LPS, CFA-I and Flagellin (Sears et al., 2017).
The antibodies produced in IMM-124E have been found to have a stronger binding and neutralizing activity (than the antibodies of unvaccinated cattle) against a wide range of LPS antigens including both the variable O-polysaccharide region and the preserved oligosaccharide core 'R' region of LPS from the 13 serotypes used in the ETEC vaccine.
IMM-124E is manufactured into a tablet form referred to as Travelan-
IMM-529
Immuron is developing IMM-529 as an adjunctive therapy in combination with standard of care antibiotics for the prevention and/or treatment of recurrent Clostridioides difficile infection (CDI). IMM-529 antibodies targeting Clostridioides difficile (C. diff) may help to clear CDI infection and promote a quicker re-establishment of normal gut flora, providing an attractive oral preventative for recurrent CDI.
Immuron is collaborating with Dr. Dena Lyras and her team at Monash University, Australia to develop vaccines to produce bovine colostrum-derived antibodies. Dairy cows were immunised to generate hyperimmune bovine colostrum (HBC) that contains antibodies targeting three essential C. diff virulence components. IMM-529 targets Toxin B (TcB), the spores and the surface layer proteins of the vegetative cells.
This unique 3-target approach has yielded promising results in pre-clinical infection and relapse models, including (1) Prevention of primary disease (80% P =0.0052); (2) Protection of disease recurrence (67%, P <0.01) and (3) Treatment of primary disease (78.6%, P<0.0001; TcB HBC). Importantly IMM-529 antibodies cross-react with whole cell lysates of many different human strains of C. diff including hypervirulent strains.
To our knowledge, IMM-529 is, to date, the only investigational drug that has shown therapeutic potential in all three phases of the disease (Hutton et al., 2017).
ProIBS
Immuron has an exclusive distribution agreement with Calmino goup AB for the territories of Australia and New Zealand for ProIBS. ProIBS is a certified medical device for the treatment of IBS symptoms such as abdominal pain, bloating and unsettled bowel movements (diarrhoea and/or constipation). ProIBS contains AVH200, derived from the plant Aloe barbadensis. Mill. AVH200 has gel forming components which support the intestinal mucosal barrier. As IBS is known to affect individuals for a long period of time, it is essential to have a treatment appropriate for long-term use -as ProIBS is. The product is safe, and no interactions with other medications are known. Science-driven innovative Calmino group AB, the developer of ProIBS, conducted a usability study among 1,003 users. PROIBS was helpful for 94% of them. 91% of the users experienced an improvement in daily life and 98% would recommend PROIBS to someone else. To learn more please check: www.proibs.eu.
Irritable bowel syndrome (IBS) is a common condition where you experience symptoms related to your digestive system. This is sometimes linked to certain foods, lifestyle habits and stress levels or mood. IBS affects around 3 out of every 10 people. Females are more likely than males to be affected. Some key symptoms of IBS include: abdominal pain or discomfort; stomach bloating and wind; chronic diarrhoea or constipation, or alternating between the two (healthdirect.gov.au). According to available data, the IBS treatment market in Australia is estimated to be a part of the broader "Digestives & Intestinal Remedies" market, generating a revenue of around AU$221.14 million in 2025, with a projected annual growth rate of 3.28% (Statista).
References
Connor P, Porter CK, Swierczewski B and Riddle MS. Diarrhea during military deployment: current concepts and future directions. Curr Opin Infect Dis. 25(5): 546-54; 2012.
Hutton, M.L., Cunningham, B.A., Mackin, K.E. et al. Bovine antibodies targeting primary and recurrent Clostridium difficile disease are a potent antibiotic alternative. Sci Rep 7, 3665 (2017). https://doi.org/10.1038/s41598-017-03982-5
Leung AK, Robson WL, Davies HD. Travelers' diarrhea. Adv Ther. Jul-Aug; 23(4): 519-27; 2006
Otto W, Najnigier B, Stelmasiak T and Robins-Browne RM. Randomized control trials using a tablet formulation of hyperimmune bovine colostrum to prevent diarrhea caused by enterotoxigenic Escherichia coli in volunteers Scandinavian Journal of Gastroenterology 46: 862- 868; 2011.
Riddle MS, Sanders JW, Putnam SD, and Tribble DR. Incidence, etiology, and impact of diarrhea among long-term travelers' (US military and similar populations): A systematic review. American Journal of Tropical Medicine and Hygiene. 74(5): 891-900; 2006.
Sears KT, Tennant SM, Reymann MK, Simon R, Konstantopolos N, Blackwelder WC, Barry EM and Pasetti MF. Bioactive Immune Components of Anti-Diarrheagenic Enterotoxigenic Escherichia coli Hyperimmune Bovine Colostrum products. Clinical and Vaccine Immunology. 24 (8) 1-14; 2017.
Steffen R. Epidemiology of travelers' diarrhea. J Travel Med. 24(suppl_1): S2-S5; 2017.
For more information visit: https://www.immuron.com.au/ and https://www.travelan.com
Sign up to Immuron's Investor Hub: Here
FORWARD-LOOKING STATEMENTS:
This press release may contain "forward-looking statements" within the meaning of Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934, each as amended. Such statements include, but are not limited to, any statements relating to our growth strategy and product development programs and any other statements that are not historical facts. Forward-looking statements are based on management's current expectations and are subject to risks and uncertainties that could negatively affect our business, operating results, financial condition and stock value. Factors that could cause actual results to differ materially from those currently anticipated include: risks relating to our growth strategy; our ability to obtain, perform under and maintain financing and strategic agreements and relationships; risks relating to the results of research and development activities; risks relating to the timing of starting and completing clinical trials; uncertainties relating to preclinical and clinical testing; our dependence on third-party suppliers; our ability to attract, integrate and retain key personnel; the early stage of products under development; our need for substantial additional funds; government regulation; patent and intellectual property matters; competition; as well as other risks described in our SEC filings. We expressly disclaim any obligation or undertaking to release publicly any updates or revisions to any forward-looking statements contained herein to reflect any change in our expectations or any changes in events, conditions or circumstances on which any such statement is based, except as required by law.
COMPANY CONTACT:
Steven Lydeamore
Chief Executive Officer
steve@immuron.com