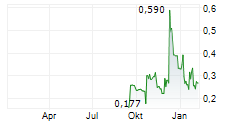
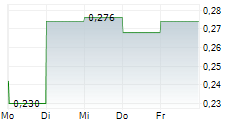
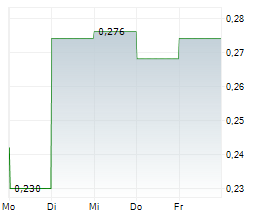
Vancouver, British Columbia--(Newsfile Corp. - February 26, 2026) - DiagnosTear Technologies Inc. (CSE: DTR) (FSE: X8F) ("DiagnosTear" or the "Company"), a fast-growing innovator in point-of-care ocular diagnostics, is pleased to announce that it has obtained all required regulatory and ethics approvals to proceed with the clinical study of TeaRx Red Eye previously announced in a press release dated December 2, 2025.
As previously disclosed, the Company initiated a collaboration with AlyaTec, a leading Contract Research Organization (CRO) specializing in allergy and environmental diseases, to conduct a clinical study designed to advance the analytical validation and FDA submission pathway of the TeaRx Red Eye diagnostic platform.
The Company has now received:
- Approval from ANSM (Agence Nationale de Sécurité du Médicament et des Produits de Santé), the French National Agency for Medicines and Health Products Safety.
- A favourable opinion from the French Ethics Committee (Comité de Protection des Personnes Sud Méditerranée III), which reviewed the study protocol and issued a positive decision following its plenary session.
These approvals enable the clinical study to formally proceed.
In addition, AlyaTec has notified the Company that operational readiness has been achieved and that patient recruitment and sampling has commenced.
This study is designed to evaluate tear samples collected from documented allergy patients before and after controlled ocular allergy provocation, with the goal of finalizing the analytical validation of total tear IgE as a key biomarker for Allergic Conjunctivitis and further strengthening the scientific and regulatory pathway of TeaRx Red Eye, as previously described. The significance of a successful outcome in this study would be validation of the ability of the TeaRx Red Eye test to identify active ocular allergy by total tear IgE.
Dr. Shimon Gross, CEO of DiagnosTear, commented: "Obtaining both regulatory and ethics approvals represents a significant milestone for our Red Eye program and reflects the strong scientific and clinical foundation of this study. With recruitment now underway, we are entering an important execution phase that moves us closer to completing the analytical validation of our multi-biomarker diagnostic platform."
Dr. Gross added: "We are pleased to see the rapid operational progress at AlyaTec and the initiation of patient screening. We believe these developments meaningfully advance our regulatory strategy and support our broader commercialization roadmap."
About DiagnosTear Technologies Inc.
DiagnosTear Technologies Inc. is a global leader in rapid, point-of-care diagnostics for ocular diseases. The Company develops multi-parametric tests that provide fast, clinically actionable insights based on tear fluid analysis. DiagnosTear's mission is to transform ophthalmic care with accessible diagnostics that support precise, data-driven decision making.
About AlyaTec
AlyaTec is a French Contract Research Organization (CRO) specializing in allergy research. Known for its advanced clinical models and translational expertise, AlyaTec supports companies worldwide in advancing innovative diagnostic and therapeutic solutions.
On Behalf of the Board of Directors
Dr. Shimon Gross, CEO
For further information, please contact:
Dr. Shimon Gross
Chief Executive Officer
DiagnosTear Technologies Inc.
+972-52-3408550
shimon@diagnostear.com
www.diagnostear.com
THE CANADIAN SECURITIES EXCHANGE HAS NOT REVIEWED AND DOES NOT ACCEPT RESPONSIBILITY FOR THE ACCURACY OR ADEQUACY OF THIS RELEASE, NOR HAS OR DOES THE CSE'S REGULATION SERVICES PROVIDER.
Forward-Looking Statements
This news release contains "forward-looking information" within the meaning of applicable Canadian securities legislation. Forward-looking information includes, but is not limited to, statements regarding the anticipated clinical study with AlyaTec, the expected timeline and results of patient recruitment and sampling, the analytical validation of TeaRx Red Eye and total tear IgE as a biomarker for Allergic Conjunctivitis, the Company's regulatory strategy including its FDA submission pathway, and the Company's broader commercialization roadmap. Forward-looking information is based on certain assumptions and expectations of the Company, including that the clinical study will proceed as planned, that patient recruitment will be completed in a timely manner, that study results will support the analytical validation of the TeaRx Red Eye platform, and that regulatory submissions will advance on the anticipated timeline.
Forward-looking information is subject to known and unknown risks, uncertainties and other factors that may cause the actual results, level of activity, performance or achievements of the Company to be materially different from those expressed or implied by such forward-looking information, including but not limited to: risks related to clinical study outcomes; regulatory risks and uncertainties; risks related to the Company's ability to obtain additional financing; competition; changes in general economic conditions; and other risks described in the Company's public disclosure documents filed on SEDAR+ at www.sedarplus.ca. Although the Company has attempted to identify important factors that could cause actual results to differ materially from those contained in forward-looking information, there may be other factors that cause results not to be as anticipated, estimated or intended.
There can be no assurance that such information will prove to be accurate, as actual results and future events could differ materially from those anticipated in such information. The Company does not undertake to update any forward-looking information, except in accordance with applicable securities laws. Readers should not place undue reliance on forward-looking information.

To view the source version of this press release, please visit https://www.newsfilecorp.com/release/285470
Source: DiagnosTear Technologies Inc.