Oak Ridge, TN, March 03, 2026 (GLOBE NEWSWIRE) -- Daxor Corporation (Nasdaq: DXR), the global leader in blood volume measurement technology today issued a corporate update in a Letter to Shareholders filed in its certified shareholder report on Form N-CSR on March 2, 2026 from CEO & President, Michael Feldschuh.
Dear Valued Shareholders,
"To measure is to know. If you cannot measure it, you cannot improve it." - Lord Kelvin
Lord Kelvin's timeless scientific principle perfectly encapsulates the mission of Daxor Corporation. In medicine, you cannot effectively treat what you cannot accurately measure. Looking back at the entirety of 2025, our commitment to replacing clinical guesswork with precise measurement - changing healthcare outcomes and economics - has culminated in a truly transformative year for Daxor Corporation. Last year marked a crucial inflection point for our company, characterized by groundbreaking product innovation, rapidly expanding market adoption, undeniable clinical validation, and increased visibility for our Blood Volume Analysis (BVA) technology.
Financial and Operating Performance Highlights
We are particularly pleased to report on our financial performance for the year ended December 31, 2025, demonstrating significant progress and continued growth in our operating business.
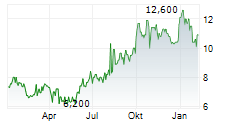
As of December 31, 2025, Daxor's net assets were $45,887,266, reflecting an increase of $10,094,965 from $35,792,301 at December 31, 2024. On a per-share basis, our Net Asset Value increased to $9.07 per share at December 31, 2025, up $1.82 per share from $7.25 per share at December 31, 2024.
For the year ended December 31, 2025, Daxor had net dividend income of $13,598 and net realized gains on investment activity of $745,334. There was a net decrease in unrealized appreciation on investments of $684,006, as positions were sold during 2025 and prior periods' significant unrealized gains unwound into realized gains. The Net Increase in Net Assets Resulting From Operations of $9,169,774 included non-cash stock-based compensation expense of $765,906.
Importantly, our operating division continued to show remarkable growth throughout the year. The operating division experienced a 45 percent increase in unaudited revenues for the year ended December 31, 2025, as compared to the year ended December 31, 2024. This substantial growth was driven by a combination of sales of our single-use blood volume diagnostic kits for heart failure management and critical care use, as well as revenue from military contracts. The addition of new accounts during this period also contributed to this revenue growth.
Additionally, the operating division realized a loss of $268,598 for the entire year of operations due to judicious investment in research and development for the 2025 product launch, ramping commercial sales teams, and production facilities for our next-generation analyzers. This is significantly lower than the $1,614,545 realized loss from the same period of the prior year, demonstrating the substantial operational growth of the division over the last year in our drive to a break-even operating performance despite the significant investments required to hit key milestones in development.
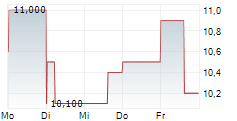
To further bolster our balance sheet and support this growth, subsequent to year-end, we successfully closed a $9 million registered direct offering in January 2026, priced at $11.75 per share. This capital injection significantly strengthens our working capital and provides the resources needed to further accelerate our commercialization efforts as we expand our market footprint, but our 2025 year-end numbers do not reflect this infusion of capital.
A Milestone Year: FDA Clearance & Commercial Momentum
Our most significant corporate highlight of the year was the receipt of FDA 510(k) clearance in August for our next-generation rapid, compact, portable Blood Volume Analyzer. This new system, developed under contract with the U.S. Department of Defense, changes the game for fluid management. It provides critical results three times faster than our BVA-100 system, weighs just seven pounds, and can be easily moved between settings,all while delivering the same highly accurate, laboratory-grade diagnostic precision.
The market has responded enthusiastically. We are utilizing our newly raised capital to assemble next generation units for placement, while engaging in the expansion of our sales team. Our growth is being strongly propelled by our dual-solution strategy, offering both our on-site BVA analyzers for immediate assessment and our CLIA-certified ezBVA Lab Service with 24-hour turnaround results. In late 2025, we announced accelerating market adoption with expansions into an Upper Midwest Integrated Health System, an Ohio Academic Medical Center, and a Southwest Cardiology Group Practice. Throughout the broader year, we also secured new adoptions across Kentucky, Arkansas, Philadelphia, and Florida. To celebrate our FDA clearance and expanding market presence, our team proudly rang the Nasdaq Closing Bell in August. We also actively engaged with the broader medical and investment communities, showcasing our technology at the MedAxiom Cardiovascular Transforum, the ISHLT Annual Meeting, and the H.C. Wainwright 27th Annual Global Investment Conference, among others.
Unprecedented Clinical Validation
The clinical validation supporting our BVA technology reached new heights in 2025, further proving that accurate, real-time blood volume measurement is essential for value-based care:
- Critical Care & Sepsis: In September, a landmark pilot study co-authored by leading intensive care experts was published in the Journal of Critical Care. The study revealed pervasive misdiagnoses using traditional clinical assessments, noting that 48% of COVID-19 patients were clinically deemed hypervolemic when objective measures showed they were hypovolemic. The study highlighted the urgent need for reliable tools like BVA to prevent insufficient fluid administration or dangerous fluid overload in the ICU
- Heart Failure Survival: New research presented at ACC25 revealed that Daxor BVA-identified euvolemic heart failure patients experienced 2.61 times better survival-a crucial finding underscoring the life-saving potential of accurate blood volume management
- Prestigious Publications: A significant Duke University study on Blood Volume Analysis in Heart Failure was published in the American Heart Journal in May, further solidifying the scientific evidence backing our technology's utility in managing complex cardiovascular conditions
Corporate Structure & Strategic Realignment
On February 10, 2026, Daxor Corporation formally announced its intention to transition its primary regulatory reporting framework from the Investment Company Act of 1940 back to the Securities Exchange Act of 1934. We anticipate completing the necessary regulatory steps to fully effectuate this change no later than the end of the second quarter if not sooner.
During the month of December 2025, Daxor Corporation sold the remaining positions of the investment portfolio and currently does not hold positions in common or preferred stocks. Although the time and resources dedicated to the investment activities of Daxor Corporation have been very limited over the last few years, our time and resources are now 100% dedicated to the growth of what has been our primary business, bringing Blood Volume to those who need it to optimize treatment.
This strategic transition to the Exchange Act is a profound financial validation of our focused growth strategy. It aligns our regulatory profile with our true identity as an innovative, high-growth medical diagnostic company. Furthermore, reporting under the Exchange Act will simplify our financial disclosures, provide investors with standardized GAAP metrics directly comparable to other publicly traded medical diagnostic companies, improve transparency, and garner deeper institutional interest and analyst coverage.
In summary, our mission is to advance patient care through precise blood volume management, solving one of the greatest challenges in healthcare. I thank you for your continued trust and investment in Daxor Corporation. We look forward to sharing further updates as we continue our mission.
Sincerely,
Michael Feldschuh President and CEO Daxor Corporation
Any shareholder who is interested in learning more about our medical instrumentation and biotechnology operations should visit our website at www.daxor.com or contact our investor relations representative Bret Shapiro of CORE IR at www.coreir.com for more detailed information. We periodically issue press releases regarding research reports and placements of the Daxor BVA system in hospitals. Sign up to receive news on Daxor's innovative technology and investor announcements HERE.
About Daxor Corporation
Daxor Corporation (Nasdaq: DXR) is tackling healthcare's "multi-billion-dollar silent crisis", the inability to precisely measure blood volume. This often results in suboptimal care, prolonged hospital stays, and increased readmissions for many high-cost medical conditions like heart failure and those requiring ICU care. With 50 years of experience and innovation, Daxor is proud to manufacture and distribute its patented, FDA-cleared Blood Volume Analysis (BVA) diagnostic which offers unmatched, real-time, precise data via its rapid, hand-held, lab-based system. This empowers clinicians to make individualized treatment decisions that significantly improve patient outcomes and deliver substantial efficiencies in value-based healthcare. Daxor is ISO certified and operates a U.S.-based, 20,000-square-foot state-of-the-art manufacturing facility, positioning the company for accelerated market expansion.
Forward-Looking Statements
Certain statements in this release may include forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, including without limitation statements regarding the impact of hiring sales staff and expansion of our distribution channels. Forward-looking statements are predictions, projections and other statements about future events that are based on current expectations and assumptions and, as a result, are subject to risks and uncertainties. Many factors could cause actual future events to differ materially from the forward-looking statements in this release, including, without limitation, those risks associated with our post-market clinical data collection activities, benefits of our products to patients, our expectations with respect to product development and commercialization efforts, our ability to increase market and physician acceptance of our products, potentially competitive product offerings, intellectual property protection, FDA regulatory actions, our ability to integrate acquired businesses, our expectations regarding anticipated synergies with and benefits from acquired businesses, and additional other risks and uncertainties described in our filings with the SEC. Forward-looking statements speak only as of the date when made. Daxor does not assume any obligation to publicly update or revise any forward-looking statements, whether as a result of new information, future events or otherwise.
Investor Relations Contact
Bret Shapiro
COO - Head of Capital Markets
COREIR
516-222-2560
brets@coreir.com|www.coreir.com