Endospan, a privately-held company pioneering solutions for the endovascular repair of aortic arch disease, today announced that the U.S. Food and Drug Administration (FDA) has approved the NEXUS Aortic Arch Stent Graft.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260407530235/en/
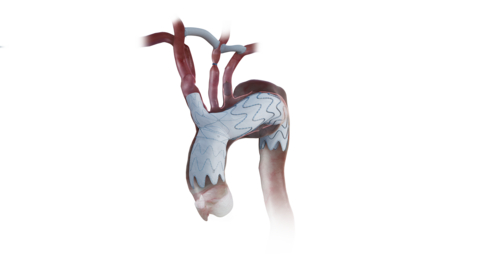
NEXUS Aortic Arch Stent Graft
FDA approval was supported by the one-year results of the TRIOMPHE Investigational Device Exemption (IDE) Study evaluating the NEXUS System shown to effectively treat aortic arch disease, including chronic aortic dissections, in patients at high risk for open surgical repair. The approval marks a critical milestone for the Company and enables a US commercial launch of the NEXUS System.
The NEXUS System is a bimodular system designed to mimic the ascending and arch anatomy. It has the lowest profile 20F delivery system with a pre-shaped catheter to allow one pass into the arch to minimize arch manipulation and an integrated branch designed for hemodynamic efficiency.
TRIOMPHE is a prospective, multicenter study designed to assess the safety and effectiveness of the NEXUS System in high-risk surgical patients. The one-year results for the high-risk surgical patients with chronic dissections demonstrated safe and effective treatment in the ascending aorta, a very difficult segment of the aorta to treat without high rates of co-morbidities such as mortality or stroke.
"The anatomical design of the NEXUS System addresses many of the complexities that occur when treating the ascending and aortic arch," said Brad Leshnower, national cardiac surgery co-principal investigator. "I am pleased to now be able to offer this minimally invasive solution to patients with life threatening diseases involving the ascending aorta and arch who are at high risk for conventional open repair."
More than 120,000 patients suffer from thoracic aortic arch disease every year in the U.S. and Europe, with only about 25% diagnosed or treated. Despite significant advancements, patients with excessive risk or anatomical factors might not be indicated for surgery, yet anatomical complexity and lack of approved devices for the arch have often prohibited endovascular repair. This makes the choice of treatment difficult or even impossible for some patients. Providing the alternative of transcatheter repair expands the treatment options for the aortic community.
"On behalf of my co-Principal National Investigator Dr. Leshnower and me, I would like to thank the entire TRIOMPHE investigational body for their commitment, time and effort to make this technology available and expand our treatment options for delivering aortic care to the community," said Dr. Ross Milner, Louis Block Professor of Surgery, Chief, Section of Vascular Surgery and Endovascular Therapy.
"This is an exciting milestone for our team, and we are grateful for the dedication and partnership of all the physicians and clinical staff involved in the TRIOMPHE Study," said Kevin Mayberry, CEO. "NEXUS was designed specifically for the ascending and aortic arch. The TRIOMPHE one-year data and pre-and-post commercialization data from the EU demonstrate that this is a viable alternative for patients with arch disease and we are pleased, with the FDA approval, to be able to offer NEXUS to U.S. patients."
About Endospan
Privately held Endospan, headquartered in Herzliya (Tel Aviv), Israel, is a pioneer in the endovascular repair of aortic arch disease including aneurysms and dissections. Endospan's NEXUS Aortic Arch Stent-Graft System was the ?rst endovascular off-the-shelf system with CE Mark to treat an underserved group of patients diagnosed with a dilative lesion in, or near, the aortic arch and ascending expanding minimally invasive endovascular repair in this anatomical region. For additional information about Endospan, visit its website at www.endospan.com.
View source version on businesswire.com: https://www.businesswire.com/news/home/20260407530235/en/
Contacts:
Kevin Mayberry, CEO
kevin@endospan.com
