KATY, TX / ACCESS Newswire / April 30, 2026 / A groundbreaking study published in the prestigious journal Cell Metabolism (IF=30.9) has revealed a novel signaling axis connecting gut microbiota, immune cells, and progenitor adipocytes, offering a fresh target for metabolic disease intervention. The study demonstrates that cold exposure significantly increases the levels of Prevotella copri and its metabolic product, 3-phenylpropionic acid (3-PPA), which activates communication between immune cells and progenitor cells to promote beige adipogenesis. This thermogenic process helps combat obesity.
The research highlights the crucial role of high-quality recombinant proteins in unraveling complex biological mechanisms, with Wuhan-based Cloud-Clone providing the key tool for validating the findings through its recombinant CXCL13 protein.

A New Metabolic Pathway: From Gut to Fat
How does the gut microbiota influence our energy expenditure? This study offers an exciting new answer.
The research team first confirmed that depleting gut microbiota impairs cold-induced beige fat generation. However, colonizing the gut with Prevotella copri restored the thermogenic effect. Metabolomics analysis revealed that P. copri efficiently produces a small molecule metabolite-3-phenylpropionic acid (3-PPA).
Supplementing with 3-PPA mimicked the metabolic benefits of P. copri colonization: it promoted the browning of inguinal white adipose tissue (iWAT), enhancing the thermogenic capacity and energy expenditure of mice, without affecting the normal function of classic brown adipose tissue (BAT).
In simpler terms, 3-PPA acts like a "thermogenesis amplifier," where cold exposure acts as the switch, amplifying the signal that turns white fat into heat-generating beige fat.
Targeting Not Fat Cells, But "Fat Stem Cells"
Interestingly, 3-PPA does not directly act on mature adipocytes but rather targets SMA? adipocyte progenitor cells (APCs), driving their differentiation into beige fat cells. Lineage tracing experiments confirmed that SMA? progenitor cells are the primary source of beige fat induced by 3-PPA.
In intervention experiments, supplementing with 3-PPA in a cold environment significantly prevented obesity and metabolic disorders induced by a high-fat diet. This effect strictly depended on cold exposure, underscoring that 3-PPA acts as an amplifier rather than an independent trigger. Moreover, this mechanism was verified in human adipocyte progenitor cells and porcine models, demonstrating its potential for cross-species translation.
CXCL13: The Immune Bridge Connecting Metabolites to Fat Thermogenesis
How does 3-PPA activate adipocyte progenitors? The mechanistic analysis led to an unexpected discovery: 3-PPA does not directly affect progenitor cells but first activates a subset of M2-like macrophages. The process unfolds as follows:
3-PPA binds to the FFAR1 receptor on macrophages, enhancing their oxidative phosphorylation metabolism.
This induces the secretion of the chemokine CXCL13, which recruits CXCR5? follicular helper T cells (Tfh) to white adipose tissue.
Tfh cells secrete IL-4, activating the STAT6 signaling pathway in SMA? progenitor cells, promoting beige adipogenesis.
This immune-progenitor cell crosstalk forms an intricate "immune-stem cell" network, where M2-like macrophages, Tfh cells, and SMA? progenitor cells collaborate to create an immune microenvironment conducive to beige fat formation. CXCL13 plays a central role in connecting microbial metabolites with progenitor cell activation.
The Power of Precision Tools: Recombinant CXCL13 in Mechanistic Insights
To clarify CXCL13's role in beige fat generation, the research team utilized highly active recombinant CXCL13 (rCXCL13) for a series of validations. Results showed that, under standard differentiation conditions, rCXCL13 did not directly upregulate UCP1 expression in beige adipocytes-confirming that CXCL13 does not directly act on adipocytes and providing a critical basis for downstream mechanistic investigations.
Further experiments revealed the following:
rCXCL13 significantly increased CXCR5 expression in iWAT lymph nodes.
Knockdown of Cxcr5 with lentivirus completely blocked rCXCL13-induced UCP1 upregulation, confirming CXCR5 as the essential receptor.
RNA-seq and flow cytometry analyses confirmed that both rCXCL13 and 3-PPA selectively upregulated Tfh cell markers and increased CD4? Tfh cells in iWAT.
Co-culture experiments provided decisive evidence: rCXCL13 promoted beige fat formation only in the presence of Tfh cells. Tfh cell depletion nullified the effect.
Using rCXCL13, the team mapped out a complete signaling chain: CXCL13 - Tfh cells - IL-4 - beige fat generation.
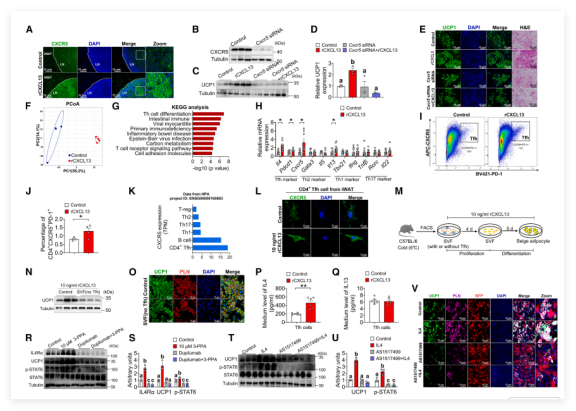
Figure 1: CXCL13 recruits and activates Tfh cells through the IL-4/STAT6 signaling pathway, promoting the formation of beige adipocytes (Image from Cell Metabolism)
Insights and Implications: Dual Value in Mechanistic Discovery and Tool Application
This study not only uncovers a new mechanism by which 3-PPA regulates beige fat formation through immune-progenitor cell dialogue but also highlights the essential role of high-quality recombinant proteins in deciphering complex physiological processes. The rigorous application of rCXCL13 enabled the team to distinguish between direct and indirect effects, identify target cell populations, and confirm the signaling hierarchy.
For researchers in metabolism, immunology, and microbiome studies, selecting high-quality, validated recombinant proteins is critical to the depth of mechanistic exploration and the reliability of conclusions.
Cloud-Clone: High-Quality Recombinant Proteins for Scientific Validation and Translational Applications
In the validation of CXCL13-mediated beige fat generation, Wuhan-based Cloud-Clone's recombinant CXCL13 protein (RPB601Mu01) played a pivotal role. The research team relied on this highly active protein to accurately map the CXCL13?Tfh cells?IL-4?beige fat generation signaling axis, establishing CXCL13 as the core immune mediator linking microbial metabolites to thermogenesis.
Cloud-Clone offers:
Over 17,000 recombinant proteins, covering multiple species including humans, mice, rats, and pigs.
Protein expression, purification, and small molecule antigen modification platforms, enabling large-scale production via bioreactors to meet industrial demands.
More than 1,000 active proteins, including cytokines (TGF-ß1, TNF-a, IL-2), growth factors (GM-CSF, EGF), enzymes (peroxidase, acetyl heparinase, DNA polymerase), and IVD targets (AMH, PINP).
Strict activity validation: All proteins undergo multiple biological assays to ensure stability and reliability for basic research and translational applications.
Whether exploring cutting-edge mechanisms or developing diagnostic reagents and therapeutics, Cloud-Clone is committed to providing researchers worldwide with high-confidence, batch-consistent recombinant proteins.
Reference
Cell Metab. 2026 Apr 7;38(4):763-778.e7.
DOI: 10.1016/j.cmet.2026.01.017. Epub 2026 Feb 24.
Microbial-derived 3-phenylpropionic acid orchestrates immune-progenitor cell crosstalk to promote beige adipogenesis and energy expenditure.
About Cloud-Clone Corp.
Cloud-Clone Corp. is dedicated to the development and production of high-quality immunoassay reagents and detection solutions. With a focus on antibody engineering, multiplex assay development, and cross-platform compatibility, the company provides research tools designed to support precision medicine and advanced biomedical investigation globally.
Our core products and services include the research and development of proteins, antibodies, ELISA kits, primary cells, and multiplex assay kits, as well as professional CRO services to fully meet the diverse needs of biomedical research and related fields.
For more information about Cloud-Clone Corp, visit www.cloud-clone.com.
CLOUD-CLONE CORP.(CCC)
Tel: 001-832-538-0970, 0086-27-8425-9552
Email: mail@cloud-clone.com, sales@cloud-clone.us
Visit us on social media:
LinkedIn: https://www.linkedin.com/company/cloud-clone-corp/
Instagram: https://www.instagram.com/cloudclonecorp2023/
TickTok: https://www.tiktok.com/@cloudclonecorp
SOURCE: Cloud-Clone Corp. (CCC)
View the original press release on ACCESS Newswire:
https://www.accessnewswire.com/newsroom/en/biotechnology/cloud-clone-supports-groundbreaking-cell-metabolism-study-gut-microbial-metabolite-pr-1162825



