Issuer: Immunic AG
/ Key word(s): Quarter Results
Immunic, Inc. Reports First Quarter 2026 Financial Results and Provides Corporate Update - Appointed Globally Renowned Biopharmaceutical Executive and Neurology Drug Developer, Michael A. Panzara, M.D., M.P.H., as Chief Medical Officer - - Continued to Execute Phase 3 ENSURE Trials of Vidofludimus Calcium in Relapsing Multiple Sclerosis, with Top-Line Data Expected by End of 2026 - - Raised $200 Million in an Oversubscribed Private Placement, with Potential for up to an Additional $200 Million - NEW YORK, May 13, 2026 - Immunic, Inc. (Nasdaq: IMUX), a late-stage biotechnology company pioneering the development of novel oral therapies for neurologic diseases, today announced financial results for the first quarter ended March 31, 2026, and provided a corporate update. "We are fast approaching a highly pivotal juncture, with the anticipated top-line data readout of the twin phase 3 ENSURE trials of our lead asset, orally available nuclear receptor-related 1 (Nurr1) activator, vidofludimus calcium (IMU-838), in relapsing multiple sclerosis (RMS), expected by the end of 2026," stated Daniel Vitt, Ph.D., Chief Executive Officer of Immunic. "In anticipation, we strengthened our leadership team with the recent appointment of Dr. Michael A. Panzara as Chief Medical Officer. Mike brings deep expertise in neurology and a proven track record of advancing multiple sclerosis (MS) therapies through late-stage clinical development and global regulatory approvals. Additionally, we enhanced our Board of Directors with the appointment of Jon Congleton, who has nearly four decades of biopharmaceutical leadership experience. Earlier in the quarter, Simona Skerjanec, M.Pharm, M.B.A., who joined Immunic's Board of Directors in July 2024, has been elevated to interim Chairperson of the Board and Thor Nagel, Principal at BVF Partners L.P., has been appointed as a member of the Board. Together, these key appointments are intended to best position the company for successful execution of our late-stage development priorities and prepare for potential commercialization." Dr. Vitt continued, "As important, in February, we closed an oversubscribed private placement financing of up to $400 million in gross proceeds, with $200 million received upfront. This highly successful transaction signals investors' continued confidence in Immunic and has provided the resources we need to advance our programs through key milestones and to continue our transition into a commercial-stage company. The initial proceeds are expected to fund our operations through the completion of our phase 3 ENSURE trials in RMS and our planned RMS New Drug Application (NDA) submission in the United States in mid-2027. The funds will also support continued investment in our development organization and launch readiness. At the same time, we remain focused on expanding the opportunity for vidofludimus calcium beyond RMS, which is supported by the growing body of data from our phase 2 CALLIPER trial in progressive MS (PMS) and our plan to initiate a confirmatory phase 3 program in primary progressive MS (PPMS) later this year." Jason Tardio, President and Chief Operating Officer of Immunic, added, "We believe vidofludimus calcium has the potential to offer a transformative approach to disease modification in MS. Unlike currently available oral therapies that primarily target inflammatory pathways, vidofludimus calcium is designed to deliver both direct neuroprotective effects through Nurr1 activation and anti-inflammatory activity via selective DHODH inhibition. In clinical trials to date, vidofludimus calcium has demonstrated a favorable safety and tolerability profile. Taken together, these attributes may support a compelling benefit-risk profile in the global MS market, which is projected to exceed $30 billion by the early 2030s." First Quarter 2026 and Subsequent Highlights
Anticipated Clinical Milestones
Financial and Operating Results
About Immunic, Inc. Immunic, Inc. (Nasdaq: IMUX) is a late-stage biotechnology company pioneering the development of novel oral therapies for neurologic diseases. The company's lead development program, vidofludimus calcium (IMU-838), is currently in phase 3 clinical trials for the treatment of relapsing multiple sclerosis, for which top-line data is expected to be available by the end of 2026. It has already shown therapeutic activity in phase 2 clinical trials in relapsing-remitting multiple sclerosis, progressive multiple sclerosis and other diseases. Vidofludimus calcium combines neuroprotective effects, through its mechanism as a first-in-class nuclear receptor-related 1 (Nurr1) activator, with additional anti-inflammatory and anti-viral effects, by selectively inhibiting the enzyme dihydroorotate dehydrogenase (DHODH). The company's development pipeline also includes earlier-stage programs, including IMU-856 and IMU-381, aimed at building a broader therapeutics platform addressing neurodegenerative, chronic inflammatory, and autoimmune-related diseases. For further information, please visit: www.imux.com. Cautionary Statement Regarding Forward-Looking Statements This press release contains "forward-looking statements" that involve substantial risks and uncertainties for purposes of the safe harbor provided by the Private Securities Litigation Reform Act of 1995. All statements, other than statements of historical facts, included in this press release regarding strategy, future operations, future financial position, future revenue, projected expenses, sufficiency of cash and cash runway, expected timing, development and results of clinical trials, prospects, plans and objectives of management are forward-looking statements. Examples of such statements include, but are not limited to, statements relating to Immunic's development programs and the targeted diseases; the potential for Immunic's development programs to safely and effectively target diseases; preclinical and clinical data for Immunic's development programs; the feasibility of advancing vidofludimus calcium to a confirmatory phase 3 clinical trial in progressive multiple sclerosis; the timing of current and future clinical trials, anticipated clinical milestones and regulatory approvals; the nature, strategy and focus of the company and further updates with respect thereto; the development and commercial potential of any product candidates of the company; expectations regarding the capitalization, resources and ownership structure of the company; and the executive and board structure of the company. Immunic may not actually achieve the plans, carry out the intentions or meet the expectations or projections disclosed in the forward-looking statements and you should not place undue reliance on these forward-looking statements. Such statements are based on management's current expectations and involve substantial risks and uncertainties. Actual results and performance could differ materially from those projected in the forward-looking statements as a result of many factors, including, without limitation, increasing inflation, tariffs and macroeconomics trends, impacts of the Ukraine - Russia conflict and the conflict in the Middle East on planned and ongoing clinical trials, risks and uncertainties associated with the ability to project future cash utilization and reserves needed for contingent future liabilities and business operations, the availability of sufficient financial and other resources to meet business objectives and operational requirements, and the ability to raise sufficient capital to continue as a going concern, the fact that the results of earlier preclinical studies and clinical trials may not be predictive of future clinical trial results, any changes to the size of the target markets for the company's products or product candidates, the protection and market exclusivity provided by Immunic's intellectual property, risks related to the drug development and the regulatory approval process and the impact of competitive products and technological changes. A further list and descriptions of these risks, uncertainties and other factors can be found in the section captioned "Risk Factors," in the company's Annual Report on Form 10-K for the fiscal year ended December 31, 2025, filed with the SEC on February 26, 2026, and in the company's subsequent filings with the SEC. Copies of these filings are available online at www.sec.gov or ir.imux.com/sec-filings. Any forward-looking statement made in this release speaks only as of the date of this release. Immunic disclaims any intent or obligation to update these forward-looking statements to reflect events or circumstances that exist after the date on which they were made. Immunic expressly disclaims all liability in respect to actions taken or not taken based on any or all of the contents of this press release. Contact Information US IR Contact US Media Contact
Dissemination of a Corporate News, transmitted by EQS News - a service of EQS Group. |
Aktien»Nachrichten»IMMUNIC AKTIE»EQS-News: Immunic AG: Immunic, Inc. Reports First Quarter 2026 Financial Results and Provides Corporate Update
| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
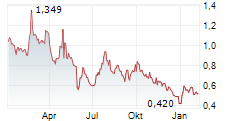
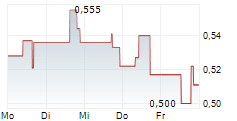
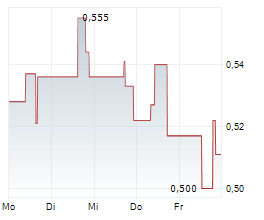
| 12,320 | 12,695 | 09:56 | |
| 12,460 | 12,805 | 02.06. |
EQS-News: Immunic AG: Immunic, Inc. Reports First Quarter 2026 Financial Results and Provides Corporate Update
© 2026 EQS Group
| Zeit | Aktuelle Nachrichten | ||
|---|---|---|---|
| Do | Immunic Therapeutics: Ehemaliger Roche-Manager Lundgren neuer CEO | ||
| Do | EQS-News: Immunic AG: Immunic to Present Additional Phase 2 CALLIPER Trial Data for Vidofludimus Calcium at the CMSC Annual Meeting 2026, Reinforcing Its Potential in Progressive Multiple Sclerosis | Issuer: Immunic AG
/ Key word(s): Conference/Study results
Immunic to Present Additional Phase 2 CALLIPER Trial Data for Vidofludimus Calcium at the CMSC Annual Meeting 2026,... ► Artikel lesen | |
| 27.05. | Immunic Therapeutics Welcomes New CEO Erik Lundgren | ||
| 27.05. | Immunic: Stifel bestätigt "Buy"-Rating nach CEO-Wechsel vor wichtigen Studiendaten | ||
| 27.05. | Immunic stock holds at Buy as Stifel cites new CEO ahead of trial |
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Werbehinweise: Die Billigung des Basisprospekts durch die BaFin ist nicht als ihre Befürwortung der angebotenen Wertpapiere zu verstehen.
Wir empfehlen Interessenten und potenziellen Anlegern den Basisprospekt und die Endgültigen Bedingungen zu lesen,
bevor sie eine Anlageentscheidung treffen, um sich möglichst umfassend zu informieren, insbesondere über die potenziellen
Risiken und Chancen des Wertpapiers. Sie sind im Begriff, ein Produkt zu erwerben, das nicht einfach ist und schwer zu
verstehen sein kann.