Foralumab is the first intranasal immune modulator in clinical trials for patients with Multiple System Atrophy
BOSTON, May 14, 2026 (GLOBE NEWSWIRE) -- Tiziana Life Sciences, Ltd. (Nasdaq: TLSA) ("Tiziana"), a biotechnology company developing its lead candidate, intranasal foralumab, a fully human, anti-CD3 monoclonal antibody, announces initial quantitative PET imaging results from the first two patients with Multiple System Atrophy (MSA) in the Phase 2 clinical trial treated with intranasal foralumab. This is the third indication that has shown a marked reduction in inflammation on PET scans following treatment with intranasal foralumab.
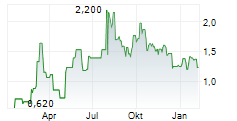
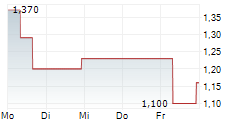
Quantitative analysis of PET scans demonstrated reductions in inflammatory activity in clinically relevant brain regions known to be affected in MSA following treatment with intranasal foralumab. In the first two treated patients, investigators observed up to approximately 35% reduction in standardized uptake value (SUV) and approximately 24% reduction in standardized uptake value ratio (SUVR) in affected areas of the brain.
Figure 1. 2nd MSA patient on nasal foralumab
Significant reduction in basal ganglia and thalamic uptake following nasal foralumab
Figure 2. 2nd MSA patient on nasal foralumab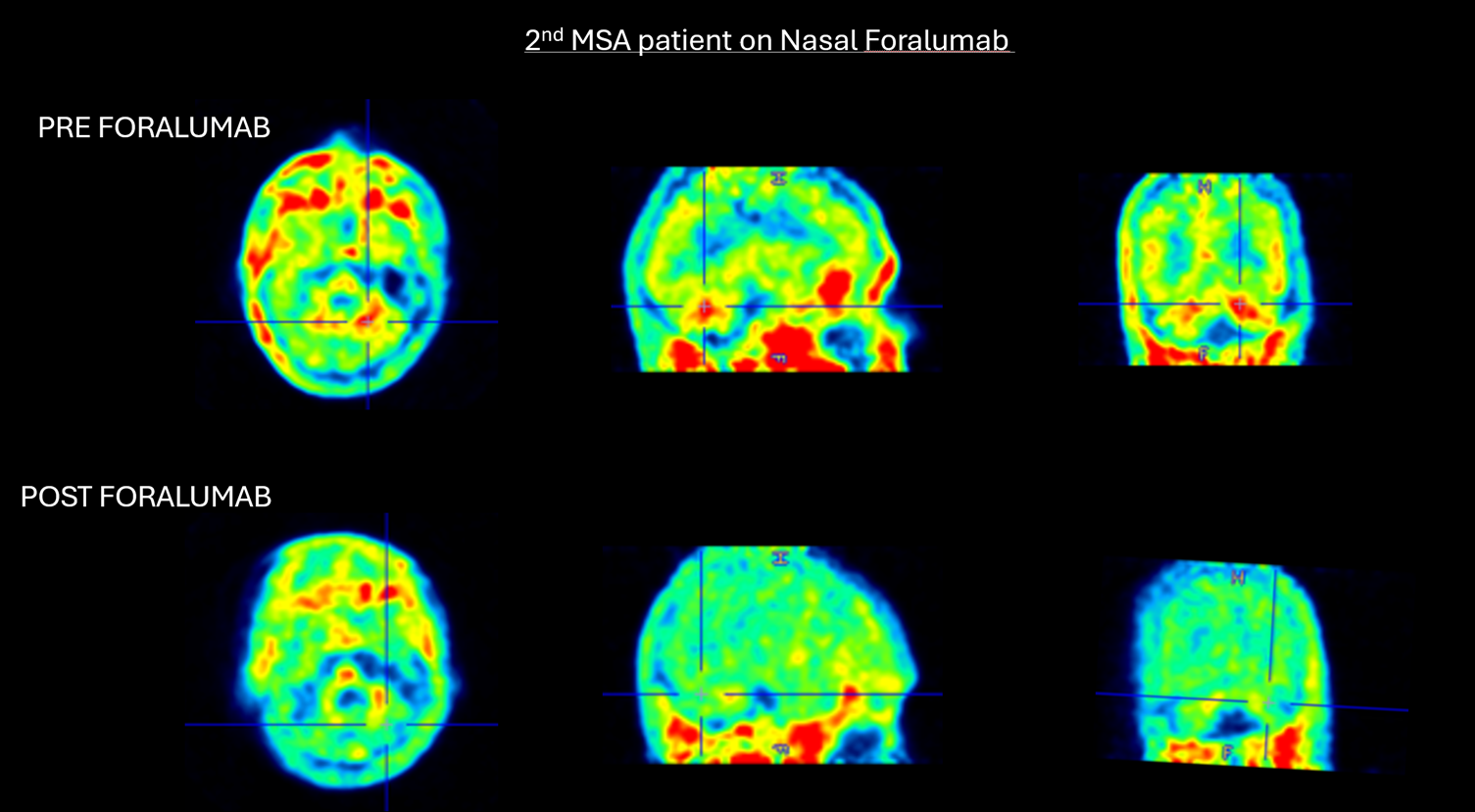
Marked reduction in Cerebellar white matter uptake following nasal foralumab
Figure 3. 1st MSA patient on nasal foralumab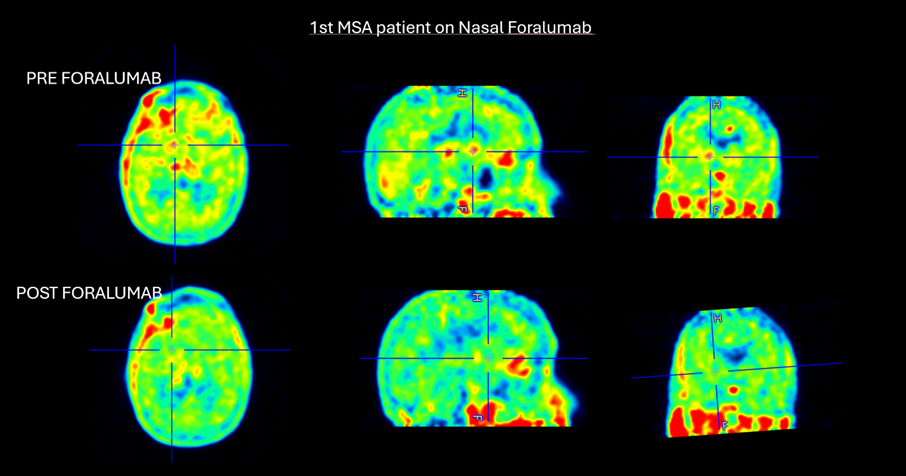
Marked reduction in basal ganglia uptake following nasal foralumab
The reductions were observed in regions including the basal ganglia and cerebellar white matter, which are known to be involved in the neurodegenerative processes underlying MSA.
"These early PET imaging findings provide quantitative evidence supporting the biological activity of intranasal foralumab in patients with MSA," said Tarun Singhal, MBBS, M.D., Founding Director, NeuroPET Program, Ann Romney Center for Neurologic Diseases, Brigham and Women's Hospital, Mass General Brigham. "In the first two MSA patients we evaluated, we observed up to approximately 35% reduction in standardized uptake value and a 24% reduction in SUV ratio in the affected brain regions. We focused on the most severely affected, clinically relevant areas known to be involved in MSA. Based on the available data, these appear to be robust quantitative findings and are comparable to results previously observed in patients with multiple sclerosis treated with intranasal foralumab, although there are methodological differences."
"This is the third indication that we have seen a marked reduction in inflammation on PET scans following treatment with intranasal foralumab," said Ivor Elrifi, Chief Executive Officer of Tiziana Life Sciences. "Building on the encouraging results previously reported in non-active secondary progressive multiple sclerosis and moderate Alzheimer's disease, these initial MSA data further highlight the potential of our therapy to meaningfully address neuroinflammation across multiple neurodegenerative conditions with high unmet need."
PET imaging comparisons conducted before and after treatment demonstrated marked reductions in radiotracer uptake across several disease-relevant brain regions. These findings suggest a reduction in neuroinflammatory activity following administration of intranasal foralumab.
Intranasal foralumab is a fully human anti-CD3 monoclonal antibody designed to modulate the immune system through mucosal tolerance mechanisms and reduce inflammation without the systemic toxicities associated with traditional anti-CD3 therapies.
MSA is a rare, progressive neurodegenerative disorder characterized by autonomic dysfunction, parkinsonism, and cerebellar ataxia. There are currently no approved disease-modifying treatments for the condition.
Tiziana plans to continue evaluating intranasal foralumab in additional patients to further validate these early findings and better characterize the therapy's potential to reduce neuroinflammation in MSA.
About Foralumab
Foralumab, a fully human anti-CD3 monoclonal antibody, is a biologic candidate that has been shown to stimulate T regulatory cells when dosed intranasally. Currently, 14 patients with Non-Active Secondary Progressive Multiple Sclerosis (na-SPMS) have been dosed in an open-label intermediate sized Expanded Access (EA) Program (NCT06802328) with either an improvement or stability of disease seen within 6 months in all patients. In addition, intranasal foralumab is currently being studied in a Phase 2a, randomized, double-blind, placebo-controlled, multicenter, dose-ranging trial in patients with non-active secondary progressive multiple sclerosis (NCT06292923).
Foralumab is the only fully human anti-CD3 monoclonal antibody (mAb) currently in clinical development. Immunomodulation by intranasal foralumab represents a novel avenue for the treatment of neuroinflammatory and neurodegenerative human diseases.[1],[2],[3]
About Tiziana Life Sciences
Tiziana is a clinical-stage biopharmaceutical company developing breakthrough therapies using transformational drug delivery technologies to enable alternative routes of immunotherapy. Tiziana's innovative nasal approach has the potential to provide an improvement in efficacy as well as safety and tolerability compared to intravenous (IV) delivery. Tiziana's lead candidate, intranasal foralumab, which is the only fully human anti-CD3 mAb currently in clinical development, has demonstrated a favorable safety profile and clinical response in patients in studies to date. Tiziana's technology for alternative routes of immunotherapy has been patented with several applications pending and is expected to allow for broad pipeline applications.
For more information about Tiziana and its innovative pipeline of therapies, please visit www.tizianalifesciences.com.
Forward-Looking Statements
Certain statements made in this announcement are forward-looking statements. These forward-looking statements are not historical facts but rather are based on the Tiziana's current expectations, estimates, and projections about its industry, its beliefs, and assumptions. Words such as 'anticipates,' 'expects,' 'intends,' 'plans,' 'believes,' 'seeks,' 'estimates,' and similar expressions are intended to identify forward-looking statements. These statements are not guarantees of future performance and are subject to known and unknown risks, uncertainties, and other factors, some of which are beyond the Tiziana's control, are difficult to predict, and could cause actual results to differ materially from those expressed or forecasted in the forward-looking statements. Tiziana cautions security holders and prospective security holders not to place undue reliance on these forward-looking statements, which reflect the view of Tiziana only as of the date of this announcement. Actual results may differ materially from those indicated by such forward-looking statements as a result of various important factors, including: the uncertainties related to market conditions and other factors described more fully in the section entitled 'Risk Factors' in Tiziana's Annual Report on Form 20-F for the year ended December 31, 2025, and other periodic reports filed with the Securities and Exchange Commission. The forward-looking statements made in this announcement relate only to events as of the date on which the statements are made. Tiziana will not undertake any obligation to release publicly any revisions or updates to these forward-looking statements to reflect events, circumstances, or unanticipated events occurring after the date of this announcement except as required by law or by any appropriate regulatory authority.
For further inquiries:
Tiziana Life Sciences Ltd
Paul Spencer, Business Development, and Investor Relations
+44 (0) 207 495 2379
email: info@tizianalifesciences.com
[1] https://www.pnas.org/doi/10.1073/pnas.2220272120
[2] https://www.pnas.org/doi/10.1073/pnas.2309221120
[3] https://www.neurology.org/doi/10.1212/NXI.0000000000200543
Photos accompanying this announcement are available at:
https://www.globenewswire.com/NewsRoom/AttachmentNg/99d6ad08-520b-4053-84a7-6a378f6a1aca
https://www.globenewswire.com/NewsRoom/AttachmentNg/83d9b57c-86b4-4051-9cb7-ebf80d6722d3
https://www.globenewswire.com/NewsRoom/AttachmentNg/82c2bf70-ed4a-4ea3-859b-65f97f65bad2