Sandoz Group AG
/ Key word(s): Regulatory Approval
MEDIA RELEASE
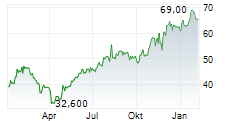
Basel, 15 May 2026 - Sandoz (SIX:SDZ/OTCQX:SDZNY), the global leader in affordable medicines, today confirms that the European Commission has granted marketing authorisation for Bysumlog (insulin lispro) and Dazparda (insulin aspart). The two biosimilar insulins, for injection in prefilled pens, are developed by Gan & Lee Pharmaceuticals.
End of Media Release |
| Language: | English |
| Company: | Sandoz Group AG |
| Centralbahnstrasse 4 | |
| 4051 Basel | |
| Switzerland | |
| Internet: | www.sandoz.com |
| ISIN: | CH1243598427 |
| Valor: | 124359842 |
| Listed: | SIX Swiss Exchange |
| EQS News ID: | 2321852 |
| End of News | EQS News Service |
2321852 15.05.2026 CET/CEST