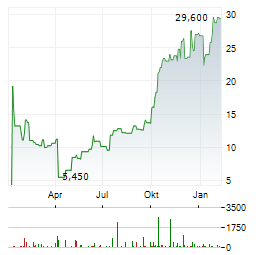
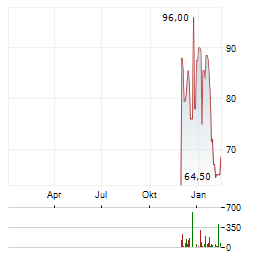
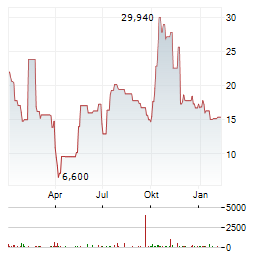
Chart-Typ:
Zeitraum:
| XETR NEW INSTRUMENTS AVAILABLE ON XETRA - 04.06.2026 | Folgende Wertpapiere werden in den Handel an folgenden Handelsplätzen (MIC Code) im Quotation Board an der Frankfurter Wertpapierbörse (FWB) aufgenommen. Die Notierung an anderen Handelsplätzen im Quotation... ► Artikel lesen |
| Oruka Therapeutics, Inc.: Oruka Therapeutics Reports First Quarter 2026 Financial Results and Provides Corporate Update | ORKA-001 EVERLAST-A 16-week data showed 63.5% PASI 100 rate and a favorable tolerability profile, with longer-term data expected in 2H 2026 ORCA-SURGE Phase 2 trial of ORKA-002 in psoriasis initiated... ► Artikel lesen |
| Halozyme Therapeutics, Inc.: Halozyme and Oruka Announce Global Collaboration and License Agreement to Develop ORKA-001 with Hypercon Technology | Agreement provides exclusive rights for IL-23p19 in psoriatic disease and option for one additional target SAN DIEGO and MENLO PARK, Calif., May 6, 2026 /PRNewswire/ -- Halozyme Therapeutics... ► Artikel lesen |
| Palvella Therapeutics Inc.: Palvella Therapeutics Announces Completion of FDA Pre-NDA Meeting for QTORIN Rapamycin in Microcystic Lymphatic Malformations | Palvella remains on track to submit an NDA in the second half of 2026 Microcystic lymphatic malformations are serious, chronic, lifelong vascular malformations that can cause persistent leaking, bleeding... ► Artikel lesen |
| Palvella Therapeutics Inc.: Palvella Therapeutics Strengthens QTORIN Pitavastatin Intellectual Property with Yale-Licensed U.S. Patent Providing Protection into 2043 | Yale-licensed U.S. patent provides broad protection for topical mevalonate pathway inhibition in porokeratosis, including disseminated superficial actinic porokeratosis (DSAP) DSAP is a serious, rare... ► Artikel lesen |
| Palvella Therapeutics Inc.: Palvella Therapeutics Announces New Data from the Phase 2 TOIVA Trial of QTORIN Rapamycin in Cutaneous Venous Malformations Presented at the 83rd Annual Meeting of the Society for Investigative Dermatology | 100% of patients with bleeding at baseline demonstrated a statistically significant improvement on the Cutaneous Venous Malformations Investigator Global Assessment Bleeding scale (cVM-IGA Bleeding)... ► Artikel lesen |
| Morning Market Movers: Neurogene, Duolingo, Xponential Fitness, Emergent BioSolutions See Big Swings | WASHINGTON (dpa-AFX) - At 6:22 a.m. ET on Friday, premarket trading is seeing notable activity in several stocks, with early price movements signaling potential opportunities before the opening... ► Artikel lesen |
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu PARDES BIOSCIENCES INC finden Sie auf Wallstreet Online.