| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
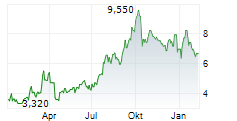
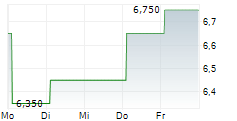
| 5,950 | 6,250 | 21:02 |
Chart-Typ:
Zeitraum:
| Qiagen erhöht Dividende und stellt weitere Aktienrückkäufe in Aussicht | DJ Qiagen erhöht Dividende und stellt weitere Aktienrückkäufe in Aussicht
DOW JONES--Die Aktionäre von Qiagen können sich über eine höhere Dividende freuen. Wie es in der Einladung zur Hauptversammlung... ► Artikel lesen |
| QIAGEN N.V.: QIAGEN plant die Ernennung von Dr. Metin Colpan zum Ehrenvorsitzenden des Aufsichtsrats, setzt die Neuausrichtung der Aufsichtsratsstruktur fort und schlägt höhere Kapitalrückzahlungen auf der Hauptversammlung 2026 vor | Mitgründer und ehemaliger CEO Dr. Metin Colpan soll zum Ehrenvorsitzenden des Aufsichtsrats ernannt werden, nachdem er beschlossen hat, nicht mehr zur Wiederwahl anzutreten Robert McMahon... ► Artikel lesen |
| QIAGEN N.V.: QIAGEN to Appoint Dr. Metin Colpan as Honorary Chairman of the Supervisory Board, Continue Governance Renewal and Propose Increased Returns at 2026 Annual General Meeting | Co-founder and former CEO Dr. Metin Colpan to become Honorary Chairman of the Supervisory Board after deciding not to stand for re-election Robert McMahon nominated for election to Supervisory... ► Artikel lesen |
| ROUNDUP: Evotec beschafft sich frisches Geld für Umbau - Aktie unter Druck | HAMBURG (dpa-AFX) - Der Wirkstoffforscher Evotec hat sich zur Finanzierung seines weiteren Umbaus frisches Geld beschafft. Das Unternehmen platzierte am Dienstagabend Wandelanleihen im Volumen von... ► Artikel lesen |
| Evotec Aktie: Rebound voraus!? Insider kaufen, Analysten bleiben bullish! | © Foto: Adam auf Pixabay (Symbolbild)Der Hamburger Biotechkonzern Evotec hat turbulente Monate hinter sich. Der Kurs hat massiv gelitten, die Quartalszahlen enttäuschten eher, und das Unternehmen steckt... ► Artikel lesen |
| Evotec rutscht erneut ab - das ist der Grund | Kräftig nach unten geht es am heutigen Mittwoch bei der Aktie von Evotec. Mit Carl Zeiss Meditec gehört sie mit einem Verlust von mehr als acht Prozent zu den zwei größten Verlierern des Tages im Nebenwerte-Index... ► Artikel lesen |
| Arcutis Biotherapeutics, Inc.: Arcutis' ZORYVE (roflumilast) Cream Receives Strong Recommendation in American Academy of Dermatology First-Ever Pediatric Atopic Dermatitis Guidelines | Strong recommendation with high certainty of clinical evidence underscores proven efficacy, safety, and tolerability of ZORYVE cream as a steroid-free topical phosphodiesterase type 4 (PDE4) inhibitorAtopic... ► Artikel lesen |
| Arcutis Biotherapeutics, Inc.: Arcutis Announces Promotion of Mas Matsuda to Executive Vice President and Chief Legal Officer to Support Next Phase of Growth | WESTLAKE VILLAGE, Calif., March 05, 2026 (GLOBE NEWSWIRE) -- Arcutis Biotherapeutics, Inc. (Nasdaq: ARQT), a commercial-stage biopharmaceutical company focused on developing meaningful innovations... ► Artikel lesen |
| Arcutis Biotherapeutics, Inc.: Arcutis Begins Enrolling Phase 1a/1b Study Evaluating ARQ-234, a CD200R Agonist, in Healthy Volunteers and Adults With Atopic Dermatitis | Phase 1a/1b, first-in-human study to evaluate safety and tolerability for investigational ARQ-234 in healthy volunteers and adults with moderate to severe atopic dermatitisARQ-234 is a potent fusion-protein... ► Artikel lesen |
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu WUXI XDC CAYMAN INC finden Sie auf Wallstreet Online.