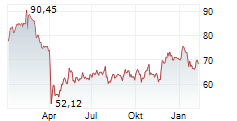
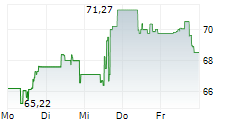
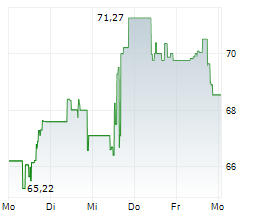
WASHINGTON (dpa-AFX) - GE HealthCare (GEHC) announced that the U.S. Food and Drug Administration has approved an updated label for its positron emission tomography or PET imaging agent Vizamyl (flutemetamol F 18 injection) for beta-amyloid detection. The revised label, effective immediately, expands the indications for use, enables quantitative analysis of Vizamyl scans, and removes significant previous limitations such as monitoring patient response to anti-amyloid therapy.
Up to now, amyloid diagnostics such as Vizamyl have been used to provide a visual assessment of amyloid plaque accumulation in the brain. With quantification now added to the label, clinicians can reach a more objective assessment, using software that enables a calculation of amyloid load, with published research demonstrating that quantification improves diagnostic confidence and consistency among readers.
In addition, with the removal of a limitation of use for monitoring therapy effectiveness, Vizamyl can also now be used to assess whether the level of amyloid plaques has been reduced sufficiently for the therapy to potentially be stopped.
Vizamyl was first approved in 2013 to estimate beta amyloid neuritic plaque density in adult patients with cognitive impairment. GE HealthCare offers solutions for quantitative analysis of amyloid PET scans including through its MIM Neuro Software platform which has been recently FDA cleared for centiloid scaling.
For More Such Health News, visit rttnews.com.
Copyright(c) 2025 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2025 AFX News