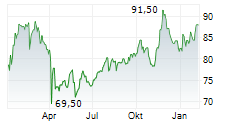
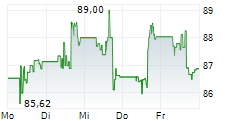
First procedures completed and enrollment underway in Embrace Gynecology study, furthering the company's progress to expand minimally invasive treatment options to patients in the United States
GALWAY, Ireland, Oct. 8, 2025 /PRNewswire/ -- Medtronic plc (NYSE: MDT) today announced the start of the Embrace Gynecology investigational device exemption (IDE) U.S. clinical study to evaluate the safety and effectiveness of its Hugo robotic-assisted surgery (RAS) system in robotic-assisted gynecological procedures.
The first procedures, total hysterectomies, were successfully completed at AHN West Penn Hospital in Pittsburgh, PA, by Dr. Sarah Crafton and Dr. Eirwen Miller, Embrace Gynecology study surgeons at Allegheny Health Network.
Embrace Gynecology is a prospective multicenter study to evaluate the safety and effectiveness of the Hugo RAS system when used during hysterectomy procedures (radical, modified radical, or total hysterectomies), inclusive of those being treated for malignancies. The study will enroll up to 70 patients across up to 5 U.S. hospitals.
"We are excited to initiate this important clinical study, which aims to investigate surgical treatment options for women in the U.S.," said Dr. Emma Rossi, Embrace Gynecology national principal investigator and associate professor of Obstetrics and Gynecology at Duke University in Durham, NC. "In my experience, women facing a gynecologic diagnosis want two things: to effectively treat their condition, and to get back to their full lives as quickly as possible. Robotic-assisted surgery helps make that possible."
The National Cancer Institute estimates that nearly 111,000 U.S. women will have been diagnosed with gynecological cancer by the end of 2025, while hundreds of thousands more will experience benign conditions such as fibroids or abnormal bleeding.1 Surgery is a common treatment for many of these gynecological conditions, and for some cancers, such as uterine (endometrial) cancer, it is usually the first and primary treatment.2 As a form of minimally invasive surgery, robotic-assisted procedures offer patients fewer complications, shorter hospital stays, and faster return to normal activities than open surgery.3.4,5
"The study name, Embrace, reflects our deeply felt compassion and care for patients and our commitment to providing access to less invasive treatment options for women," said Dr. James Porter, chief medical officer of Robotic Surgical Technologies and Digital Technologies within the Surgical business of Medtronic. "We are grateful for the strong partnership with clinical teams at our study sites and share their excitement about this rigorous scientific study that is helping to usher in a new era of choice for patients in the U.S."
Embrace Gynecology becomes the third IDE clinical study for Hugo RAS in the U.S., demonstrating Medtronic's commitment to obtaining multiple indications for its Hugo RAS system. The two other U.S. IDE clinical studies - Expand URO and Enable Hernia Repair - both met their primary safety and effectiveness endpoints.
Also, in July, positive results from a Medtronic-sponsored prospective study of the Hugo RAS system in benign gynecologic procedures outside the U.S. were shared at the Society of Robotic Surgery congress in Strasbourg, France.
The company's first U.S. submission, for a urology indication, is under review by the Food and Drug Administration and is expected later in the company's current fiscal year, followed by planned indication expansions into hernia repair and gynecology.
Born out of Medtronic's 75-year commitment to expanding access to minimally invasive surgical options worldwide in partnership with clinicians, the Hugo RAS system is currently in clinical use in more than 30 countries on five continents, with nearly 300 independent publications to date.
The Medtronic Hugo RAS system is commercially available in certain geographies. Regulatory requirements of individual countries and regions will determine approval, clearance, or market availability. In the EU, the Hugo RAS system is CE marked. In the U.S., the Hugo RAS system is an investigational device not for sale.
For more information, visit medtronic.com/hugo.
About Medtronic
Bold thinking. Bolder actions. We are Medtronic. Medtronic plc, headquartered in Galway, Ireland, is the leading global healthcare technology company that boldly attacks the most challenging health problems facing humanity by searching out and finding solutions. Our Mission - to alleviate pain, restore health, and extend life - unites a global team of 95,000+ passionate people across more than 150 countries. Our technologies and therapies treat 70 health conditions and include cardiac devices, surgical robotics, insulin pumps, surgical tools, patient monitoring systems, and more. Powered by our diverse knowledge, insatiable curiosity, and desire to help all those who need it, we deliver innovative technologies that transform the lives of two people every second, every hour, every day. Expect more from us as we empower insight-driven care, experiences that put people first, and better outcomes for our world. In everything we do, we are engineering the extraordinary. For more information on Medtronic, visit www.Medtronic.com and follow on LinkedIn.
Any forward-looking statements are subject to risks and uncertainties such as those described in Medtronic's periodic reports on file with the Securities and Exchange Commission. Actual results may differ materially from anticipated results.
Contacts:
Gary Jeanfaivre
Public Relations
+1-203-556-0777
Ryan Weispfenning
Investor Relations
+1-612-839-4549
1 https://www.aacr.org/patients-caregivers/awareness-months/gynecologic-cancer-awareness-month/
2 https://www.mayoclinic.org/diseases-conditions/endometrial-cancer/diagnosis-treatment/drc-20352466
3 Fitch K, Engel T, Bochner A. Cost differences between open and minimally invasive surgery. Managed Care. 2015;24(9):40-48.
4 Tiwari MM, Reynoso JF, High R, Tsang AW, Oleynikov D. Safety, efficacy, and cost effectiveness of common laparoscopic procedures. Surg Endosc. 2011;25(4):1127-1135.
5 Roumm AR, Pizzi L, Goldfarb NI, Cohn H. Minimally invasive: minimally reimbursed? An examination of six laparoscopic surgical procedures. Surg Innov. 2005;12(3):261-287.
SOURCE Medtronic plc