INDIANAPOLIS (dpa-AFX) - Eli Lilly and Company (LLY) said on Monday said the U.S. Food and Drug Administration (FDA) has approved a label expansion for Zepbound to include the four-dose single-patient use KwikPen which delivers a full month of treatment in one device.
Lilly added that patients opting for self-pay through LillyDirect, can receive all doses of Zepbound in a KwikPen or as single dose vials, starting at $299 per month for the 2.5 mg dose.
Zepbound is indicated for weight reduction in adults with obesity, those who are overweight with related health conditions, and for treating severe obstructive sleep apnea in adults with obesity.
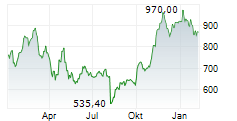
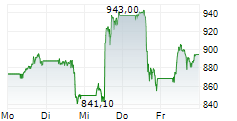
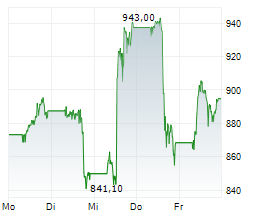
Lilly shares were trading at 1,042.11, up 3%.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News