Highlights October-December 2025
- Net sales and operating results for Q4 2025 in line with expectations: Net sales of 354 KSEK for the period (455) largely reflected royalty revenue. Operating loss was 16.7 MSEK, compared to 30.1 MSEK in the fourth quarter of 2024. Earnings per share before and after dilution were -0.03 SEK (0,02).
- Cash burn lower than guidance: Cash flow from operating activities was -19.8 MSEK, reduced from -28.3 MSEK in Q4 2024. The burn rate of 6.6 MSEK per month was well below the company's previously communicated guidance of 8 to 10 MSEK per month. Savings resulted from lower spending on clinical studies and commercial staffing expenses. Cash and cash equivalents at the end of the period equaled 77.5 MSEK (25.3), supported by a rights issue of approximately SEK 100 million before deduction of transaction costs.
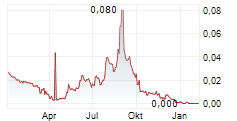
- Capital raise of 100 MSEK: On October 23, Immunovia announced the outcome of the rights issue. A total of 293,632,417 shares were subscribed for with and without subscription rights, corresponding to approximately 87.9 percent of the rights issue. The remaining 40,276,397 shares (12.1 percent) were allotted to guarantors, resulting in 100 percent subscription. Immunovia received gross proceeds of approximately SEK 100 million before deduction of transaction costs.
- PancreaSure test commercial momentum: An additional four high-risk surveillance centers initiated PancreaSure testing in the fourth quarter. New sites included Harvard-affiliated Beth-Israel Deaconess Medical Center, New York University Perlmutter Cancer Center, and Prisma Health Cancer Institute.
- Scientific support for PancreaSure: Immunovia's pivotal clinical validation study, CLARITI, was published in the prestigious journal Gastroenterology on October 27. The PancreaSure analytical validation and VERIFI clinical validation study were also published in the quarter. PancreaSure clinical data were presented by key opinion leaders at multiple scientific meetings, including the American College of Gastroenterology Annual Scientific Meeting.
- Lab accredited by CAP: On October 13, Immunovia's laboratory in North Carolina was awarded accreditation by the College of American Pathologists (CAP), affirming that the lab's operations meet rigorous global standards for quality, accuracy, and patient safety.
- Medicare reimbursement rate set: The Centers for Medicare & Medicaid Services (CMS) issued on November 26 a final payment determination for PancreaSure of $897. The Clinical Laboratory Fee Schedule (CLFS) establishes the payment rate that Medicare will pay for the PancreaSure test once coverage is in place.
Significant events after the period
- California regulatory approval: In January, Immunovia received regulatory approval from California to sell PancreaSure in the state. PancreaSure is now approved for sale in 47 of 50 US states.
"As we closed the year, the pancreatic cancer community embraced PancreaSure, publishing key studies, disseminating our data at leading scientific meetings, and implementing the test in their high-risk clinics."
Jeff Borcherding, CEO and President, Immunovia AB
Conference call
Immunovia will hold a webcast teleconference at 15:00 CET on February 24 with Jeff Borcherding, CEO and President.
To participate on the call, please dial one of the numbers or watch via the web link below.
Sweden: +46 (0)8 5051 0031
United Kingdom: +44 (0) 207 107 06 13
United States: +1 (1) 631 570 56 13
Link to the webcast:
https://creo-live.creomediamanager.com/a7a6ea3d-9185-41b9-a00f-fdbcad6ea46b
For further information, please contact
Jeff Borcherding, CEO
jeff.borcherding@immunovia.com
Immunovia in brief
Immunovia AB is a diagnostic company whose mission is to increase survival rates for patients with pancreatic cancer through early detection. Immunovia is focused on the development and commercialization of simple blood-based testing to detect proteins and antibodies that indicate a high-risk individual has developed pancreatic cancer. Immunovia collaborates and engages with healthcare providers, leading experts and patient advocacy groups to make its test available to individuals at increased risk for pancreatic cancer.
USA is the world's largest market for detection of pancreatic cancer. The Company estimates that in the USA, 1.8 million individuals are at high-risk for pancreatic cancer and could benefit from annual surveillance testing.
Immunovia's shares (IMMNOV) are listed on Nasdaq Stockholm.
For more information, please visit www.immunovia.com.