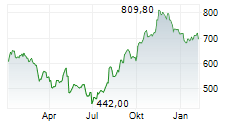
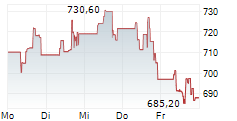
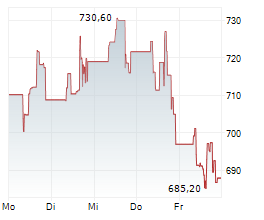
BRUSSELS (dpa-AFX) - argenx SE (ARGX) on Thursday said its Phase 3 ADAPT OCULUS study evaluating Vyvgart in adults with ocular myasthenia gravis (oMG) met its primary endpoint.
The company said patients treated with Vyvgart demonstrated a statistically significant improvement from baseline in Myasthenia Gravis Impairment Index (MGII) Patient-Reported Outcome (PRO) ocular scores at Week 4 compared with placebo. Patients receiving Vyvgart also experienced marked reductions in key ocular symptoms, including diplopia (double vision) and ptosis (drooping of the upper eyelids). Further, Vyvgart was well tolerated and showed a favorable safety profile in oMG patients, consistent with prior studies.
argenx said it expects to submit a supplemental Biologics License Application (sBLA) to the U.S. Food and Drug Administration (FDA) for Vyvgart in oMG by the end of the third quarter of 2026.
Vyvgart is already approved by the FDA for the treatment of adults with generalized myasthenia gravis, a chronic autoimmune disease.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News