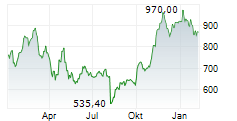
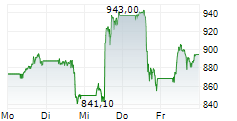
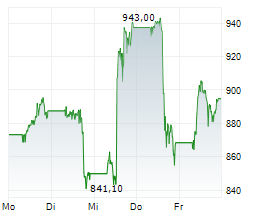
INDIANAPOLIS (dpa-AFX) - Eli Lilly and Company (LLY) on Thursday reported positive results from its Phase 3 ACHIEVE-3 trial evaluating orforglipron in adults with type 2 diabetes inadequately controlled on metformin, compared with oral semaglutide.
In the 52-week study, orforglipron outperformed semaglutide across the primary and all key secondary endpoints, showing significantly greater reductions in A1C and body weight. Specifically, orforglipron lowered A1C by 2.2%, compared with a 1.4% reduction for semaglutide.
The results were published today in The Lancet.
Lilly has submitted orforglipron to regulators in over 40 countries and plans to submit for U.S. approval for type 2 diabetes later this year.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News