WASHINGTON (dpa-AFX) - AbbVie (ABBV) on Monday announced positive results from the Phase 3 AFFIRM study evaluating subcutaneous risankizumab in moderately to severely active Crohn's disease.
At Week 12, 55 percent of patients treated with risankizumab achieved clinical remission compared to 30 percent on placebo, while 44 percent achieved endoscopic response versus 14 percent on placebo.
The benefits were observed in both treatment-naïve and refractory patients. By Week 24, following induction and maintenance therapy, 67 percent of patients achieved clinical remission and 57% endoscopic response. The safety profile was consistent with previous studies, with no new safety signals identified.
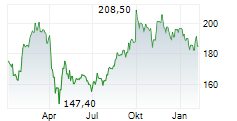
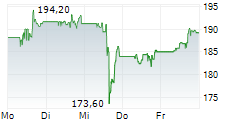
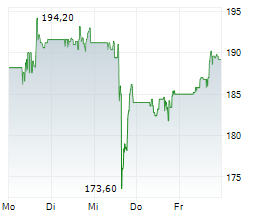
ABBV is currently trading pre market at $229.51, down $2.52 or 1.09 percent on the New York Stock Exchange.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News