Success of the transaction with €48 million raised.
Participation of leading international Healthcare specialist investors.
Funds raised are expected to support long-term value creation, primarily by expanding partnering opportunities, maximizing the value captured from future collaborations, and strengthening Medincell's proprietary long-acting injectable (LAI) technology platform through targeted innovation.
Regulatory News:
This press release is not being made in and copies of it may not be distributed or sent, directly or indirectly, into the United States, Australia, South Africa, Canada or Japan
Medincell (Euronext Paris: MEDCL), a commercial- and clinical-stage biopharmaceutical licensing company developing long-acting injectable treatments (the "Company"), today announced the successful pricing of its Private Placement (as defined below) for a total amount of €48 million through an offering to international institutional investors.
Both US and European Healthcare specialist investors, such as Perceptive Advisors, Kurma Growth Opportunities Fund, Affinity Asset Advisors and Polar Capital have participated in the transaction alongside historical main shareholders.
The net proceeds from the Private Placement, together with the Company's existing cash resources, are expected to support and accelerate the execution of Medincell's growth strategy.
In particular, the Company intends to allocate the net proceeds to:
- Expand partnering opportunities by generating high-value data through the initiation of additional programs,
- Maximize the value of future partnerships by optimizing economics and prioritizing downstream royalty participation,
- Strengthen the proprietary LAI technologyplatform through targeted innovation, including the advancement of next-generation LAI technologies that may broaden the scope, differentiation and applications of the platform.
The net proceeds may also be used for general corporate purposes, including working capital and operating expenses, and to strengthen the Company's balance sheet to support future strategic initiatives and enhance financial flexibility.
Christophe Douat, CEO of Medincell, said: "The funds raised strengthen our financial position and enable us to confidently accelerate the execution of our long-term growth strategy. We thank both our new and long-standing top-tier international investors for their continued support. This successful financing reflects their confidence in our business model, our proven ability to consistently innovate and translate innovation into value, and the significant value creation potential of our existing and future partnerships with leading global pharmaceutical companies addressing patient needs across multiple therapeutic areas."
Terms of the Private Placement
The private placement, for a total of €48 million was carried out via the issuance without shareholder's preferential subscription rights of 2,370,000 new ordinary shares, with a nominal value of 0.01 euro each, in favor of qualified investors or a restricted circle of investors referred under the provisions of article L. 411-2 1° of the French Monetary and Financial Code in accordance with the 18th resolution of the combined general meeting of the Company on September 12, 2024 (the "General Meeting") (the "Private Placement
The new shares, representing 7.1% of the share capital of the Company, on a non-diluted basis, prior to the completion of the Private Placement and 6.6% of the Company's share capital, on a non-diluted basis, following the Private Placement, were issued by decision of the Company's Chief Executive Officer (Directeur Général) dated March 5, 2026 acting under the sub delegations of authority granted by the Company's Board of Directors on March 5, 2026, in accordance with the 18th resolution of the General Meeting.
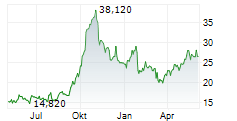
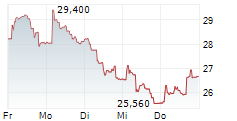
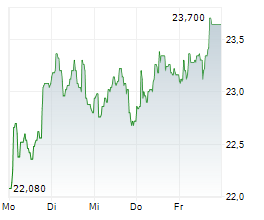
The issue price of the new shares has been set at €20.35 per share, representing a discount of 10% compared to the closing price of the Medincell share on March 5, 2026, i.e. €22.64.
By way of illustration, a shareholder holding 1.00% of the share capital of the Company prior to the launch of the Private Placement will now hold an interest of 0.93%.
To the best of the Company's knowledge, the shareholder structure before and after the completion of the Private Placement is as follows:
Medincell shareholding on a non-diluted basis | Pre-offer | Post-offer | ||
Number of shares | % of capital | Number of shares | % of capital | |
Anh Nguyen | 1,330,643 | 4.0% | 1,330,643 | 3.7% |
Sabine Hort Nguyen | 1,459,868 | 4.4% | 1,459,868 | 4.1% |
Employees and former employees and consultants | 5,344,863 | 15.9% | 5,344,863 | 14.9% |
Management Board | 1,336,569 | 4.0% | 1,336,569 | 3.7% |
Treasury shares | 899 | 0.0% | 899 | 0.0% |
Total Free float: | 24,062,174 | 71.8% | 26,432,174 | 73.6% |
o/w Mirova | 2,360,494 | 7.0% | 2,430,494 | 6.8% |
o/w Polar Capital | 1,727,475 | 5.2% | 1,962,475 | 5.5% |
o/w SITAM Belgique (Groupe Dassault) | 1,720,426 | 5.1% | 1,846,926 | 5.1% |
TOTAL | 33,535,016 | 100% | 35,905,016 | 100% |
Admission to trading of the new shares
Settlement-delivery of the new ordinary shares to be issued in the Private Placement and their admission for trading on the regulated market of Euronext Paris are expected on March 10, 2026. The new ordinary shares will be of the same category and fully fungible with the existing shares, will be entitled to all rights associated with the existing shares, and will be admitted to trading on the regulated market of Euronext Paris under the same ISIN code FR0004065605 MEDCL.
Lock-up commitments
In connection with the Private Placement, the Company, the members of the Board of Directors and certain members of the management have signed a lock-up agreement which took effect on the date of execution of the placement agreement entered into between the Company and the banks on March 5, 2026 and for a period of 90 days, subject to certain customary exceptions.
Financial Intermediaries
Jefferies, Leerink Partners, Evercore ISI are acting as Joint Global Coordinators and Joint Bookrunners and ODDO BHF SCA as Joint Bookrunner on the Private Placement. The Private Placement is subject to a placement agreement entered into between the Company and the Joint Bookrunners.
Risk factors
The attention of the public is drawn to the risk factors associated with the Company and its activity presented in Section 2 of the universal registration document filed with the French Financial Market Authority (Autorité des Marchés Financiers) (the "AMF") under number D.25-0580 on July 29, 2025, which is available free of charge on the Company's website (https://www.medincell.com/regulated-information/). The occurrence of all or part of these risks could have a negative impact on the Company's activity, financial situation, results, development or outlook. The risk factors presented in that document are the same today.
Additionally, investors are invited to consider the following risks specific to this Private Placement (i) the market price of the Company's shares may fluctuate and fall below the subscription price of the shares issued as part of the Private Placement, (ii) the volatility and liquidity of the Company's shares may fluctuate significantly, (iii) sales of the Company's shares may take place on the market and have a negative impact on the market price of its share and (iv) the Company's shareholders could suffer potentially significant dilution resulting from any future capital increases required to provide the Company with additional financing.
No Prospectus
The Private Placement is not subject to a prospectus requiring approval from the AMF.
This press release does not constitute a prospectus within the meaning of Regulation (EU) 2017/1129 of the European Parliament and of the Council of June 14, 2017, as amended, nor an offer to the public.
About Medincell
Medincell is a clinical- and commercial-stage biopharmaceutical licensing company developing long-acting injectable treatments across multiple therapeutic areas. Our innovative treatments are designed to ensure adherence to medical prescriptions, enhance the effectiveness and accessibility of medicines, and reduce their environmental impact.
These treatments combine active pharmaceutical ingredients with our proprietary BEPO BEPO Star technologies, which enables controlled drug delivery at therapeutic levels for several days, weeks, or months following a subcutaneous or local injection of a small, fully bioresorbable deposit.
Risperidone LAI was the first treatment based on BEPO technology to receive FDA approval, initially for schizophrenia in April 2023, and subsequently for Bipolar I Disorder in October 2025. It is marketed in the United States by Teva under the brand name UZEDY. Medincell's risperidone LAI was also approved for schizophrenia in Canada and South Korea in 2025.
A New Drug Application (NDA) for Olanzapine LAI as a once-monthly treatment for schizophrenia in adults was submitted to the U.S. FDA in December 2025 by Medincell's partner, Teva. U.S. FDA accepted Teva's New NDA for Olanzapine LAI on February 20, 2026.
Medincell's investigational pipeline includes numerous innovative therapeutic candidates in various stages of development, from formulation to Phase 3 clinical trials. We collaborate with leading pharmaceutical companies and foundations to advance global health through new treatment options.
Headquartered in Montpellier, France, Medincell employs over 140 people representing more than 25 nationalities.
medincell.com
UZEDY is a trademark of Teva Pharmaceuticals. Medincell's BEPO technology is licensed to Teva as SteadyTeq, a trademark of Teva Pharmaceuticals.
This announcement does not, and shall not, in any circumstances, constitute a public offering nor an invitation to the public in connection with any offer. The distribution of this document may be restricted by law in certain jurisdictions. Persons into whose possession this document comes are required to inform themselves about and to observe any such restrictions.
Not for release, directly or indirectly, in or into the United States of America, Australia, South Africa, Canada or Japan. This document (and the information contained herein) does not contain or constitute an offer of securities for sale, or solicitation of an offer to purchase securities, in the United States, Australia, South Africa, Canada or Japan or any other jurisdiction where such an offer or solicitation would be unlawful. The securities referred to herein have not been and will not be registered under the U.S. Securities Act of 1933, as amended (the "Securities Act"), and may not be offered or sold in the United States, unless the securities are registered under the Securities Act or pursuant to an exemption from the registration requirements of the Securities Act. No public offering of the securities will be made in the United States.
This communication does not constitute an offer of relevant securities to the public in the United Kingdom within the meaning of Regulation 12 of the Public Offers and Admissions to Trading Regulations 2024 (the "POATRs"), has not been approved by an authorised person in the United Kingdom for the purposes of Section 21(1) of the Financial Services and Markets Act 2000 ("FSMA") and is being distributed only to and is directed only at (a) persons outside the United Kingdom, or (b) in the United Kingdom, persons who are "qualified investors" within the meaning of paragraph 15 of Schedule 1 to the POATRs who are also (i) persons who have professional experience in matters relating to investments, falling within the meaning of Article 19(5) of the Financial Services and Markets Act 2000 (Financial Promotion) Order 2005, as amended (the "Order"), (ii) persons falling within Article 49(2)(a) to (d) of the Order (high net worth entities, unincorporated associations etc.) and (iii) persons to whom an invitation or inducement to engage in investment activity within the meaning of Section 21 of the FSMA in connection with the sale of securities may otherwise lawfully be communicated (all such persons together being referred to as "Relevant Persons"). The securities are available only to, and any invitation, offer or agreement to subscribe, purchase or otherwise acquire such securities will be available only to and will be engaged in only with, Relevant Persons. Any person who is not a Relevant Person should not act or rely on this communication or any of its contents.
In France, the offering of Medincell shares described above has been made in the context of a capital increase in favor of qualified investors or a restricted circle of investors, pursuant to Article L. 411-2 1° of the French Code monétaire et financier and applicable regulatory provisions. Pursuant to Article 211-3 of the General regulations of the AMF, Articles 1(4) and 3 of the Regulation (EU) 2017/1129 of the European Parliament and of the Council of 14 June 2017, as amended (the "Prospectus Regulation") and any applicable regulation, the offer of Medincell shares will not require the publication of a prospectus approved by the AMF.
With respect to Member States of the European Economic Area ("Member State"), no action has been taken or will be taken to permit a public offering of the securities referred to in this press release requiring the publication of a prospectus in any Member State. Therefore, such securities may not be and shall not be offered in any Member State other than in accordance with the exemptions of Article 1(4) of the Prospectus Regulation or, otherwise, in cases not requiring the publication of a prospectus under Article 3 of the Prospectus Regulation and/or the applicable regulations in such Member State.
MIFID II Product Governance/Target Market: For the sole purposes of the requirements of Article 9.8 of the EU Delegated Directive 2017/593 relating to the product approval process, the target market assessment in respect of the shares of Medincell has led to the conclusion, with respect to the type of clients criteria only that: (i) the type of clients to whom the shares are targeted is eligible counterparties and professional clients and retail clients, each as defined in Directive 2014/65/EU, as amended ("MiFID II"); and (ii) all channels for distribution of the shares of Medincell to eligible counterparties and professional clients and retail clients are appropriate. Any person subsequently offering, selling or recommending the shares of Medincell (a "distributor") should take into consideration the type of clients assessment; however, a distributor subject to MiFID II is responsible for undertaking its own target market assessment in respect of the shares of Medincell and determining appropriate distribution channels.
This press release contains forward-looking statements, including statements regarding Company's expectations for (i) the timing, progress and outcome of its clinical trials; (ii) the clinical benefits and competitive positioning of its product candidates; (iii) its ability to obtain regulatory approvals, commence commercial production and achieve market penetration and sales; (iv) its future product portfolio; (v) its future partnering arrangements; (vi) its future capital needs, capital expenditure plans and ability to obtain funding; (vii) prospective financial matters regarding our business; and (viii) use of proceeds from the private placement. Although the Company believes that its expectations are based on reasonable assumptions, any statements other than statements of historical facts that may be contained in this press release relating to future events are forward-looking statements and subject to change without notice, factors beyond the Company's control and the Company's financial capabilities.
These statements may include, but are not limited to, any statement beginning with, followed by or including words or phrases such as "objective", "believe", "anticipate", "expect", "foresee", "aim", "intend", "may", "anticipate", "estimate", "plan", "project", "will", "may", "probably", "potential", "should", "could" and other words and phrases of the same meaning or used in negative form. Forward-looking statements are subject to inherent risks and uncertainties beyond the Company's control that may, if any, cause actual results, performance, or achievements to differ materially from those anticipated or expressed explicitly or implicitly by such forward-looking statements. A list and description of these risks, contingencies and uncertainties can be found in the documents filed by the Company with the Autorité des Marchés Financiers (the "AMF") pursuant to its regulatory obligations, including the Company's universal registration document, filed with the AMF on July 29, 2025, under number D. 25-0580 (the "Universal Registration Document"), as well as in the documents and reports to be published subsequently by the Company. In particular, readers' attention is drawn to the section entitled "Facteurs de Risques" on page 30 et seq. 26 of the Universal Registration Document.
Any forward-looking statements made by or on behalf of the Company speak only as of the date they are made. Except as required by law, the Company does not undertake any obligation to publicly update these forward-looking statements or to update the reasons why actual results could differ materially from those anticipated by the forward-looking statements, including in the event that new information becomes available. The Company's update of one or more forward-looking statements does not imply that the Company will make any further updates to such forward-looking statements or other forward-looking statements. Readers are cautioned not to place undue reliance on these forward-looking statements.
This press release is for information purposes only. The information contained herein does not constitute an offer to sell or a solicitation of an offer to buy or subscribe for the Company's shares in any jurisdiction, in particular in France. Similarly, this press release does not constitute investment advice and should not be treated as such. It is not related to the investment objectives, financial situation, or specific needs of any recipient. It should not deprive the recipients of the opportunity to exercise their own judgment. All opinions expressed in this document are subject to change without notice. The distribution of this press release may be subject to legal restrictions in certain jurisdictions.
View source version on businesswire.com: https://www.businesswire.com/news/home/20260305432026/en/
Contacts:
David Heuzé
Head of Corporate and Financial Communications, and ESG
david.heuze@Medincell.com +33 (0)6 83 25 21 86
Grace Kim
Chief Strategy Officer, U.S. Finance
grace.kim@medincell.com +1 (646) 991-4023
Nicolas Mérigeau Arthur Rouillé
Media Relations
Medincell@newcap.eu +33 (0)1 44 71 94 94
Louis-Victor Delouvrier Alban Dufumier
Investor Relations France
Medincell@newcap.eu +33 (0)1 44 71 94 94