| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
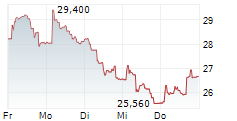
| 26,620 | 27,020 | 04.06. | |
| 26,680 | 27,080 | 04.06. |
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| MEDINCELL Aktie jetzt für 0€ handeln | |||||
| 28.05. | Medincell: Videoconference and Publication of Annual Financial Results, Tuesday, June 16, 2026 | 252 | Business Wire | Regulatory News:
Medincell (Euronext Paris: MEDCL), a commercial- and clinical-stage biopharmaceutical licensing company developing long-acting injectable treatments, today announced the hosting... ► Artikel lesen | |
| 26.05. | Medincell to Participate in Jefferies and Evercore Investor Conferences in the U.S. in June | 358 | Business Wire | Regulatory News:
Medincell (Euronext Paris: MEDCL), a commercial- and clinical-stage biopharmaceutical licensing company developing long-acting injectable treatments, today announced that Christophe... ► Artikel lesen | |
| 21.05. | EMA nimmt Zulassungsantrag von Teva und Medincell für langwirksames Schizophrenie-Medikament an | 18 | Investing.com Deutsch | ||
| 21.05. | Medincell: The European Medicines Agency Accepts Teva's Marketing Authorization Application for Olanzapine Long-Acting Injectable for the Treatment of Schizophrenia in Adults | 467 | Business Wire | The olanzapine long-acting injectable (TEV-'749 mdc-TJK) is designed to deliver the efficacy of olanzapine in a subcutaneous formulation1 administered every four weeks. If approved, TEV-'749... ► Artikel lesen | |
| 29.04. | Medincell - UZEDY: Q1 2026 Net Sales Reached $63M, Up 62% Year-on-Year | 448 | Business Wire | Regulatory News:
Medincell (Euronext Paris: MEDCL) partner, Teva Pharmaceuticals (NYSE and TASE: TEVA), announced today that UZEDY U.S. net sales reached $63 million in Q1 2026, representing a... ► Artikel lesen | |
| 22.04. | Degroof Petercam Initiates Medincell's Coverage with a "Buy" Recommendation | 546 | Business Wire | Degroof Petercam, a leading European investment bank with strong healthcare expertise, has initiated coverage of Medincell In a report entitled "Royalty flywheel approaching profitability", the... ► Artikel lesen | |
| 21.04. | Leerink Partners Initiates Medincell's Coverage with a "Outperform" Recommendation | 452 | Business Wire | Leerink Partners, a leading U.S. investment bank specialized in healthcare, has initiated coverage of Medincell The Leerink Partners equity research team has issued an "Outperform" rating on... ► Artikel lesen | |
| 16.04. | Medincell to Host an Online R&D Day on Tuesday, May 12, 2026 | 536 | Business Wire | > From Science to Long-Term Value Creation
Regulatory News:
Medincell (Euronext Paris: MEDCL) will host an online R&D Day, held in English, on Tuesday, May 12, 2026, from 4:00 to 5:30 pm CEST... ► Artikel lesen | |
| 26.03. | EIB Waives its Right to Cash Settlement on Medincell Warrants | 557 | Business Wire | The European Investment Bank (EIB) has waived the put option attached to the warrants granted in connection with the €40 million financing agreement signed in 2022 This waiver eliminates any... ► Artikel lesen | |
| 06.03. | Medincell nimmt 48 Millionen Euro über Privatplatzierung ein | 1 | Investing.com Deutsch | ||
| 06.03. | Medincell raises €48 million in private placement | 1 | Investing.com | ||
| 06.03. | Medincell Announces Successful c. €48 Million Private Placement | 522 | Business Wire | Success of the transaction with €48 million raised. Participation of leading international Healthcare specialist investors. Funds raised are expected to support long-term value creation, primarily... ► Artikel lesen | |
| 05.03. | Medincell startet Privatplatzierung über 6 % des Grundkapitals | 9 | Investing.com Deutsch | ||
| 05.03. | Medincell launches private placement for 6% of share capital | 1 | Investing.com | ||
| 05.03. | Medincell Launches a Private Placement for International Institutional Investors | 645 | Business Wire | The Private Placement, of approximately 6% of the Company's share capital, is aimed at international institutional investors, through an accelerated book-building process. Funds raised are expected... ► Artikel lesen | |
| 04.03. | Medincell to Participate in Leerink and Jefferies Miami Investor Conferences, March 8 - 11, 2026 | 530 | Business Wire | Regulatory News:
Medincell (Euronext Paris: MEDCL), a commercial- and clinical-stage biopharmaceutical licensing company developing long-acting injectable treatments, today announced its participation... ► Artikel lesen | |
| 24.02. | Medincell to Present at the TD Cowen 46th Annual Healthcare Conference in Boston, March 2-4, 2026 | 508 | Business Wire | Regulatory News:
Medincell (Euronext Paris: MEDCL), a commercial- and clinical-stage biopharmaceutical licensing company developing long-acting injectable treatments (the "Company"), today announced... ► Artikel lesen | |
| 21.02. | Teva, Medincell granted FDA review for long-acting antipsychotic | 16 | Seeking Alpha | ||
| 21.02. | FDA Accepts NDA For Teva And Medincell's Olanzapine LAI In Schizophrenia Treatment | 1.331 | AFX News | PETAH TIKVA (dpa-AFX) - Teva Pharmaceuticals, the U.S. affiliate of Teva Pharmaceutical Industries Ltd. (TEVA), together with Medincell, announced that the U.S. Food and Drug Administration... ► Artikel lesen | |
| 20.02. | Medincell: U.S. Food and Drug Administration (FDA) Accepts Teva's New Drug Application (NDA) for Olanzapine Extended-Release Injectable Suspension (TEV-'749) for the Once-Monthly Treatment of Schizophrenia in Adults | 1.012 | Business Wire | Olanzapine long-acting injectable suspension (TEV-'749) has the potential to offer the efficacy of olanzapine in a once-monthly, subcutaneous formulation 1 If approved, TEV-'749 could help... ► Artikel lesen |
1 2 3 Weiter >>
53 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BIONTECH | 78,10 | +2,49 % | BioNTech Aktie: 70 Prozent Ansprechrate! Brennt jetzt das Kursfeuerwerk ab? | © Foto: Tung Lam auf Pixabay (Symbolbild)Lange wurde BioNTech nur als Corona-Gewinner gesehen. Doch das Unternehmen schreibt gerade still und leise eine neue Geschichte, und die hat mit Pandemie kaum... ► Artikel lesen | |
| EVOTEC | 5,115 | +1,79 % | EQS-NVR: Evotec SE: Veröffentlichung der Gesamtzahl der Stimmrechte nach § 41 WpHG mit dem Ziel der europaweiten Verbreitung | EQS Gesamtstimmrechtsmitteilung: Evotec SE
/ Veröffentlichung der Gesamtzahl der Stimmrechte
Evotec SE: Veröffentlichung der Gesamtzahl der Stimmrechte nach § 41 WpHG mit dem... ► Artikel lesen | |
| MEDIGENE | 0,022 | -10,48 % | PTA-Adhoc: Medigene AG: Widerruf der Zulassung der Aktien der Medigene AG zum Handel im regulierten Markt | DJ PTA-Adhoc: Medigene AG: Widerruf der Zulassung der Aktien der Medigene AG zum Handel im regulierten Markt
Veröffentlichung von Insiderinformationen gemäß Artikel 17 MAR Schlagwort(e): Sonstiges... ► Artikel lesen | |
| QIAGEN | 32,630 | +5,34 % | DEUTSCHE BANK RESEARCH stuft QIAGEN NV auf 'Buy' | FRANKFURT (dpa-AFX Analyser) - Deutsche Bank Research hat die Einstufung für Qiagen mit einem Kursziel von 43 US-Dollar auf "Buy" belassen. Das Interesse der Investoren habe vor allem dem Tuberkulosetest... ► Artikel lesen | |
| MODERNA | 44,280 | +4,72 % | Neue CAR-T-Erfolge bei Autoimmunerkrankungen: Warum Lösungen für Zelltherapien vor dem nächsten Wachstumsschub stehen könnten | ||
| PAION | 0,100 | +23,60 % | XFRA MISTRADE ANTRAG IN ISIN DE000A3E5EG5 WURDE STATTGEGEBEN | Dem Mistrade-Antrag wurde stattgegeben. Folgende Geschaefte in der ISIN DE000A3E5EG5, Wertpapier-Name: PAION AG INH O.N., wurden aufgehoben:ISIN Datum Zeit Volumen PreisDE000A3E5EG5 02.02.2026 08:13:40 300 0... ► Artikel lesen | |
| VALNEVA | 2,479 | +2,95 % | Original-Research: Valneva SE (von First Berlin Equity Research GmbH): Buy | Original-Research: Valneva SE - von First Berlin Equity Research GmbH
28.05.2026 / 17:08 CET/CEST
Veröffentlichung einer Research, übermittelt durch EQS News - ein Service der EQS Group.
Für den... ► Artikel lesen | |
| AMGEN | 298,95 | +2,50 % | Mesoblast: Während Amgen und Novo Nordisk den Biotech-Hype treiben, arbeitet dieser Zelltherapie-Player am operativen Durchbruch | ||
| NOVAVAX | 8,760 | -0,23 % | Novavax, Inc.: Novavax Reports First Quarter 2026 Financial Results and Operational Highlights | Total revenue of $140 million in the first quarter of 2026
Pfizer agreement announced in January for non-exclusive license to utilize Matrix-M® in two infectious... ► Artikel lesen | |
| STRYKER | 257,50 | +1,18 % | Stryker launches new small bone power tool for orthopedic procedures | ||
| BIOGEN | 170,24 | +0,75 % | Biogen's Salanersen Wins FDA Breakthrough Therapy Designation For Spinal Muscular Atrophy Treatment | WESTON (dpa-AFX) - Thursday, Biogen Inc. (BIIB) announced that the U.S. Food and Drug Administration has granted salanersen Breakthrough Therapy Designation for the treatment of spinal muscular... ► Artikel lesen | |
| BIOFRONTERA | 2,490 | -4,96 % | PTA-News: Biofrontera AG: Biofrontera berichtet über die Ergebnisse des ersten Quartals 2026 | DJ PTA-News: Biofrontera AG: BIOFRONTERA BERICHTET ÜBER DIE ERGEBNISSE DES ERSTEN QUARTALS 2026
Unternehmensmitteilung für den Kapitalmarkt
Biofrontera AG: BIOFRONTERA BERICHTET ÜBER... ► Artikel lesen | |
| HEIDELBERG PHARMA | 2,710 | +1,12 % | HEIDELBERG PHARMA AG: Ruhe vor der nächsten Phase | ||
| ILLUMINA | 144,80 | -1,76 % | ILMN Stock Jumps 98.7% in a Year: What's Powering the Surge? | ||
| RECURSION PHARMACEUTICALS | 3,800 | 0,00 % | Recursion Pharmaceuticals: Recursion Reports Grant of Inducement Awards as Permitted by the Nasdaq Listing Rules |
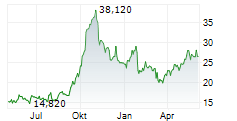
Das Unternehmen MEDINCELL SA kann der Branche Biotechnologie zugeordnet werden. Die relative Kursveränderung beträgt aktuell +4,46 % bei einem Aktienkurs von 26,700 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu MEDINCELL SA finden Sie auf Wallstreet Online.