CANBERA (dpa-AFX) - Telix Pharmaceuticals Limited (TLX, TLX.AX) announced the resubmission of its New Drug Application (NDA) to the U.S. Food and Drug Administration (FDA) for TLX101-Px (Pixclara, Floretyrosine F 18 or 18F-FET). This investigational PET imaging agent is intended to help differentiate recurrent or progressive glioma (a type of brain cancer) from treatment-related changes in both adult and pediatric patients.
The resubmission includes additional data and statistical analyses requested by the FDA. Following a Type A meeting and ongoing consultation, Telix believes the new information, combined with the original dataset, adequately addresses the FDA's Complete Response Letter.
TLX101-Px has been granted Orphan Drug and Fast Track designations, underscoring its potential to address a significant unmet medical need. Although 18F-FET PET imaging is already recognized in international clinical practice guidelines for glioma imaging, there is currently no FDA-approved targeted amino acid PET agent available in the U.S. for adult or pediatric brain cancer imaging.
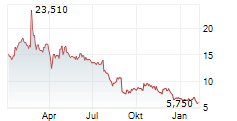
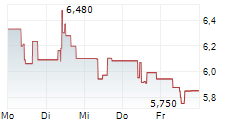
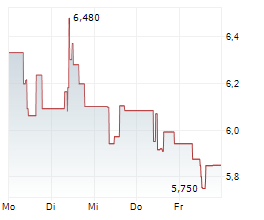
TLX.AX is currently trading at A$11.01, down A$0.28 or 2.48%.
For More Such Health News, visit rttnews.com.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News