INDIANAPOLIS (dpa-AFX) - Eli Lilly and Co. (LLY) announced Monday positive, topline results from the Phase 3 ADorable-1 trial evaluating the safety and efficacy of EBGLYSS (lebrikizumab-lbkz) in pediatric patients with moderate-to-severe atopic dermatitis.
EBGLYSS met the primary and key secondary endpoints at Week 16, improving disease severity while delivering skin clearance and relief from persistent itch.
Atopic dermatitis is more common in children than adults, affecting 9.6 million children in the U.S., one-third of whom have moderate-to-severe disease.
In the Phase 3 ADorable-1 study, 63 percent of patients achieved meaningful skin improvement (EASI-75) and 44 percent achieved clear or almost clear skin (IGA 0,1) at Week 16.
In key secondary endpoints, 39 percent of patients achieved a high bar of near-complete skin clearance (EASI-90) and 35 percent achieved significant itch relief (Pruritus NRS ?4-point improvement).
The safety and tolerability profile of EBGLYSS was consistent with adult and adolescent studies, with no injection site pain reported.
EBGLYSS is an interleukin-13 (IL-13) inhibitor that selectively blocks IL-13 signaling with high binding affinity and slow dissociation rate. The cytokine IL-13 is a primary cytokine in atopic dermatitis.
In ADorable-1, participants were randomized to receive placebo or a weight-based dose of EBGLYSS. Topical corticosteroids were required beginning two weeks before randomization and throughout the 16-week study but could be decreased or stopped once patients achieved IGA 2 or less.
The co-primary endpoints in ADorable-1 were EASI-75 and IGA 0,1 at Week 16. Key secondary endpoints included an even greater clinical improvement in disease severity (EASI-90) and itch relief.
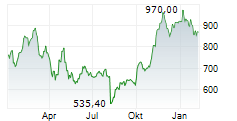
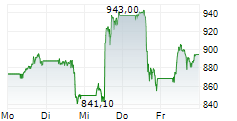
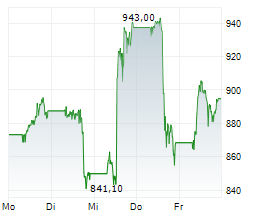
In Monday's pre-market trading, LLY is trading on the NYSE at $986.37, up $1.09 or 0.11 percent.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News