NOT FOR PUBLICATION, DISTRIBUTION OR RELEASE, DIRECTLY OR INDIRECTLY, IN WHOLE OR IN PART, IN OR INTO THE UNITED STATES OF AMERICA, AUSTRALIA, CANADA, HONG KONG, ISRAEL, JAPAN, NEW ZEALAND, SOUTH AFRICA, SWITZERLAND OR ANY OTHER JURISDICTION WHERE SUCH PUBLICATION, DISTRIBUTION OR RELEASE WOULD BE UNLAWFUL OR REQUIRE REGISTRATION OR OTHER MEASURES.
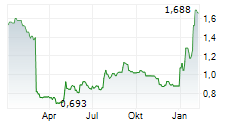
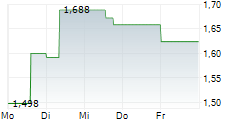
STOCKHOLM, March 23, 2026 /PRNewswire/ -- The Board of Directors of Diamyd Medical Aktiebolag ("Diamyd Medical" or the "Company") has resolved on a directed issue of 17,226,500 new Class B shares at a subscription price of SEK 13.54 per share, through which the Company receives approximately SEK 233 million before transaction costs (the "Share Issue"), and two directed issuances of an aggregate of 46,649,362 warrants (the "Warrant Issuances", and together with the Share Issue, the "Directed Issuances"), to certain U.S. sector specialist investors. Each warrant entitles the holder to subscribe for one new Class B share at an exercise price of SEK 20 per Class B share. If all warrants are exercised in full, the total gross proceeds from the Directed Issuances are expected to amount to approximately SEK 1,166 million (corresponding to approximately USD 125 million).
The Company also announces completion of randomization in the pivotal precision medicine Phase 3 DIAGNODE-3 trial evaluating retogatein (rhGAD65) in individuals with type 1 diabetes. A total of 321 patients have been randomized across 57 clinical sites in Europe and the U.S., and the study is now fully enrolled. The results from an interim analysis in the DIAGNODE-3 trial are expected to be announced on March 27, 2026, following a Data Safety Monitoring Board (DSMB) meeting.
"We are pleased to welcome leading U.S. sector specialist investors at this pivotal stage," said Ulf Hannelius, President & CEO of Diamyd Medical. "Their participation brings deep sector expertise and strong alignment with the U.S. market, which will be central to our future development and commercialization strategy. The financing strengthens our cash position, extends our anticipated runway through the full Phase 3 readout, and together with the TO 5 warrant program provides sufficient capital to support pre-commercial preparations for retogatein and continued development of our precision medicine platform."
The placement includes participation from new U.S. sector specialist investors, including Perceptive Advisors, Vestal Point Capital, RA Capital Management, Vivo Capital, Caligan Partners, Seven Fleet Capital, ADAR1 Capital Management, Great Point Partners, LLC, Logos Capital, and Sphera Healthcare.
The Board of Directors of Diamyd Medical has resolved on the Share Issue of 17,226,500 new Class B shares at a subscription price of SEK 13.54 per Class B share, corresponding to the closing price of Diamyd Medical Class B shares on Nasdaq First North Growth Market on March 20, 2026, for proceeds of approximately SEK 233 million before transaction costs. Further, the Board of Directors has resolved on the Warrant Issuances of an aggregate of 46,649,362 warrants, divided into two separate tranches, with the second tranche subject to a vote by the Company's shareholders as set out below. The warrants are issued free of charge and the exercise price corresponds to the Company's existing TO 5 warrant program (issued in connection with the rights issue dated February 28, 2025). Each warrant entitles the holder to subscribe for one new Class B share at an exercise price of SEK 20 per Class B share, which represents a premium of 47.7 percent to the closing price of Diamyd Medical Class B shares on Nasdaq First North Growth Market on March 20, 2026. The warrants may be exercised no later than 30 calendar days following the Company's public announcement of the interim efficacy analysis in its ongoing pivotal Phase 3 trial (DIAGNODE-3) evaluating retogatein (rhGAD65) in individuals with Stage 3 type 1 diabetes, however, at the latest on September 30, 2027. As set out above, the expected date for such announcement is March 27, 2026 which would mean that the last date for exercise will be April 26, 2026. If all issued warrants are exercised in full for subscription of new Class B shares, the Company will receive additional proceeds of approximately SEK 933 million.
The Company intends to use the proceeds from the Share Issue for completion of its ongoing Phase 3 DIAGNODE-3 trial, and to use the proceeds from the exercise of warrants, if any, for pre-commercial activities, life cycle management opportunities, and pipeline development. The Share Issue and 21,774,296 of the warrants issued under the Warrant Issuances have been resolved on by the Board of Directors based on the authorization granted by the annual general meeting held on December 4, 2025 ("Tranche 1"), and the remaining 24,875,066 warrants under the Warrant Issuances will be issued subject to the subsequent approval of an extraordinary general meeting, which is expected to be held on April 8, 2026 ("Tranche 2"). Chairman of the Board of Directors, Anders Essen-Möller (directly and indirectly via ARMEA Aktiebolag), Vice Chairman of the Board of Directors, Erik Nerpin, and CEO, Ulf Hannelius, together with one of the Company's principal shareholders, Bertil Lindkvist, and the other shareholding members of the founder family Essen-Möller, who as per the date hereof together hold approximately 12.8 percent of the shares and approximately 29.1 percent of the votes in the Company, have undertaken to vote in favor of Tranche 2. Notice of the extraordinary general meeting will be published through a separate press release.
The subscription price and exercise price of the Directed Issuances have been determined by the Board of Directors based on arm's length negotiations with the participating investors as well as in consultation with the Company's exclusive placement agent Leerink Partners. The Board of Directors considers the subscription price and exercise price of the Directed Issuances to be at market terms, taking into account the current market conditions. The Board of Directors of the Company deems, after an overall assessment and careful consideration, that the Directed Issuances with deviation from the shareholders' preferential rights are a better alternative for the Company's shareholders than a rights issue. A rights issue would entail significantly longer execution time and thereby increased market exposure and a higher potential risk of materially affecting the share price negatively, particularly in this volatile and challenging market. The cost of carrying out a directed issue is deemed to be lower than in a rights issue where, among other things, there would be a risk that a rights issue would not be fully subscribed. In addition, unlike a rights issue, the Directed Issuances have broadened the shareholder base and provided the Company with new reputable institutional investors, which the Board of Directors believes will strengthen the liquidity of the shares and be beneficial to the Company.
Through the Share Issue, the total number of shares will increase by 17,226,500 Class B shares, from 137,758,000 shares to 154,984,500 shares and the total number of votes by 1,722,650 votes, from 16,928,608 votes to 18,651,258 votes. The share capital will increase by SEK 1,747,133.161608, from SEK 13,971,588.545365 to SEK 15,718,721.706973. The Share Issue entails a dilution of approximately 11 percent of the number of shares and approximately 9 percent of the number of votes. If all warrants issued in the Warrant Issuances are exercised for subscription of new Class B shares in the Company, the total number of shares will increase by an additional 46,649,362 Class B shares, from 154,984,500 shares to 201,633,862 shares and the total number of votes by an additional 4,664,936.2 votes, from 18,651,258 votes to 23,316,194.2 votes. This corresponds to an additional dilutive effect from the warrants of a maximum of approximately 23 percent of the number of shares and approximately 20 percent of the number of votes, also taking into account the Class B shares issued in the Share Issue. The aggregate maximum dilution from the Directed Issuances corresponds to approximately 32 percent of the total number of shares and approximately 27 percent of the total number of votes. Settlement of Tranche 1 is expected to take place on or around March 25, 2026. Tranche 2 will be completed as soon as practically possible following the extraordinary general meeting's approval of Tranche 2, entailing that warrants are expected to be transferred to the investors on or around April 10, 2026.
In connection with the Directed Issuances, the Company has agreed to a lock-up undertaking, with customary exceptions, on future share issuances for a period of 90 days after the settlement date of Tranche 1. In addition, the members of the Board of Directors and members of the management have undertaken not to, subject to customary exceptions as well as an exception to sell shares solely to the extent necessary to fund the exercise price payable upon exercise of TO 5 warrants, divest any shares in the Company for a period of 90 days from the settlement date of Tranche 1.
Advisers
Leerink Partners acts as exclusive placement agent and G&W Fondkommission acts as co-adviser to the Company in connection with the Directed Issuances. Advokatfirman Vinge KB acts as legal adviser to the Company in connection with the Directed Issuances.
About Diamyd Medical
Diamyd Medical develops precision medicine therapies to prevent and treat type 1 diabetes. Retogatein (rhGAD65) formulated with alum is an investigational antigen-specific immunotherapy, designed to induce antigen-specific immune tolerance to GAD65 and preserve endogenous insulin production in individuals with type 1 diabetes who carry the HLA DR3-DQ2 gene. Retogatein has been granted Orphan Drug Designation in the U.S. as well as Fast Track Designation by the U.S. FDA for the treatment of Stage 3 (clinically diagnosed symptomatic) type 1 diabetes. Fast Track Designation has also been granted for the treatment of Stage 1 and 2 (pre-symptomatic) type 1 diabetes. DIAGNODE-3, a confirmatory Phase 3 trial with potential for an accelerated approval pathway in the U.S., is being conducted at 57 clinics in eight European countries and in the U.S. in patients with recent-onset (Stage 3) type 1 diabetes. Significant results in preserving endogenous insulin production have previously been shown in a large genetically predefined patient group - both in a large-scale meta-analysis as well as in the Company's prospective European Phase 2b trial. The DIAGNODE-3 trial has only included patients from this specific patient group that carries the common genotype known as HLA DR3-DQ2, which constitutes approximately 40% of patients with type 1 diabetes in Europe and the U.S. A biomanufacturing facility is under development in Umeå, Sweden, for the manufacture of retogatein (recombinant GAD65 protein), the active ingredient in the antigen-specific immunotherapy. Diamyd Medical is a major shareholder in the stem cell company NextCell Pharma AB and in the artificial intelligence company MainlyAI AB.
Diamyd Medical's Class B share is traded on Nasdaq First North Growth Market under the ticker DMYD B. FNCA Sweden AB is the Company's Certified Adviser.
CONTACT:
For further information, please contact:
Ulf Hannelius, President and CEO
Phone: +46 736 35 42 41
E-mail: ulf.hannelius@diamyd.com
Niklas Axelsson, CFO
Phone: +46 72 528 23 49
E-mail: niklas.axelsson@diamyd.com
This information was brought to you by Cision http://news.cision.com
https://news.cision.com/diamyd-medical-ab/r/diamyd-medical-enters-into-equity-financing-agreements-with-new-u-s--sector-specialist-investors-for,c4325027
The following files are available for download:
https://mb.cision.com/Main/6746/4325027/3996818.pdf | PDF version |
![]() View original content:https://www.prnewswire.co.uk/news-releases/diamyd-medical-enters-into-equity-financing-agreements-with-new-us-sector-specialist-investors-for-up-to-usd-125-million-and-announces-full-enrollment-in-phase-3-trial-302721837.html
View original content:https://www.prnewswire.co.uk/news-releases/diamyd-medical-enters-into-equity-financing-agreements-with-new-us-sector-specialist-investors-for-up-to-usd-125-million-and-announces-full-enrollment-in-phase-3-trial-302721837.html