SANTA CLARA (dpa-AFX) - Agilent Technologies (A) said it has received FDA approval for PD-L1 IHC 22C3 pharmDx, Code SK006, as a companion diagnostic to aid in identifying patients with esophageal or gastroesophageal junction carcinoma who may be eligible for treatment with KEYTRUDA, Merck's anti-PD-1 therapy. The approval marks the eighth FDA approved companion diagnostic indication currently available for PD-L1 IHC 22C3 pharmDx, Code SK006, for use with KEYTRUDA.
'With the expanded FDA approval of PD-L1 IHC 22C3 pharmDx in esophageal or GEJ carcinoma, Agilent is proud to support clinicians in identifying patients eligible for treatment with KEYTRUDA,' said Nina Green, vice-president and general manager of Agilent's Clinical Diagnostics Division.
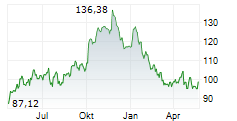
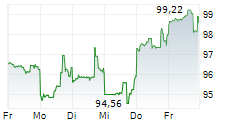
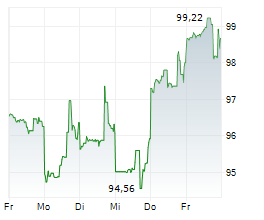
In pre-market trading on the NYSE, Agilent shares are up 0.46 percent to $113.48.
For More Such Health News, visit rttnews.com.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News