INDIANAPOLIS (dpa-AFX) - Eli Lilly and Company (LLY), Friday announced that its new long-term data of EBGLYSS study showed durable skin clearance and relief from persistent itch for up to four years for patients with moderate-to-severe atopic dermatitis.
During the open-label extension study, 75% of patients achieved a high bar of near-complete skin clearance, and 78% experienced significant itch relief.
Notably, the safety of EBGLYSS in the first year of the ADlong study was consistent with the known profile in patients with moderate-to-severe atopic dermatitis, regardless of dose frequency, and no new safety signals were observed.
The company holds the exclusive rights for development and commercialization of EBGLYSS in the U.S. and the rest of the world outside Europe. Meanwhile, its partner Almirall has licensed the rights to develop and commercialize EBGLYSS for the treatment of dermatology indications, including atopic dermatitis, in Europe.
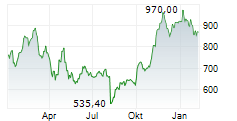
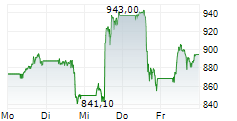
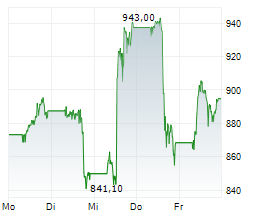
In the pre-market hours, LLY is trading at $894.77, down 0.21 percent on the New York Stock Exchange.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News