WASHINGTON (dpa-AFX) - Boston Scientific Corp. (BSX) announced that the CHAMPION-AF global clinical trial successfully met all primary and secondary safety and efficacy endpoints. The study compared the WATCHMAN FLX Left Atrial Appendage Closure (LAAC) Device with non-vitamin K antagonist oral anticoagulants (NOACs) as a first-line therapy for stroke risk reduction in patients with non-valvular atrial fibrillation (NVAF).
Key findings revealed that the WATCHMAN FLX device provided statistically superior protection from bleeding while demonstrating similar efficacy to blood thinners in reducing stroke risk among NVAF patients.
The results were presented as a late-breaking clinical trial at the American College of Cardiology's Annual Scientific Session & Expo and were simultaneously published in The New England Journal of Medicine, underscoring the potential of WATCHMAN FLX as a safe and effective alternative to long-term anticoagulant therapy.
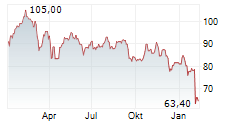
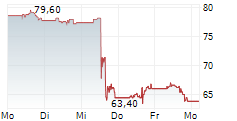
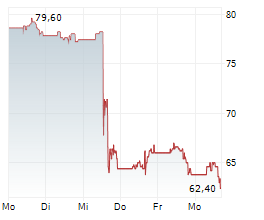
BSX closed Friday's regular trading at $69.17 down $1.00 or 1.43%.
For More Such Health News, visit rttnews.com.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News