WASHINGTON (dpa-AFX) - Waters (WAT) announced that the FDA has cleared the Onclarity HPV Self-Collection Kit and approved the BD Onclarity HPV Assay with extended genotyping for at-home use. Waters said it is establishing partnerships to enable nationwide access to the Onclarity HPV Self-Collection Kit, which is expected to be available by prescription in the coming months.
The kit is tested with the BD Onclarity HPV Assay, which is the only FDA-approved HPV assay to identify six individually and three groups of pooled results. Samples are processed on the fully automated BD COR System.
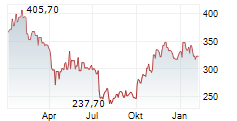
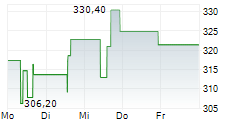
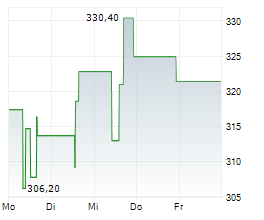
In pre-market trading on NYSE, Waters shares are up 2 percent to $313.53.
For More Such Health News, visit rttnews.com.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News