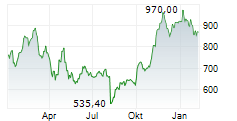
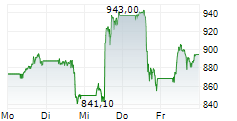
INDIANAPOLIS (dpa-AFX) - Eli Lilly and Company (LLY) on Monday announced positive topline results from the Phase 3 BRUIN CLL-322 study evaluating Jaypirca in combination with venetoclax and rituximab in patients with relapsed or refractory chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL).
The study met its primary endpoint, showing that adding Jaypirca to venetoclax and rituximab significantly improved progression-free survival compared with the standard combination, as assessed by an independent review committee. The benefit was consistent across key patient subgroups, including those previously treated with a covalent BTK inhibitor.
The safety profile of the combination was in line with the known safety of the individual medicines.
Lilly said it plans to submit the data to regulators later this year to seek an expansion of Jaypirca's label.
Jaypirca is currently approved for treating adult patients with relapsed or refractory CLL or SLL who have previously received a covalent BTK inhibitor, as well as for relapsed or refractory mantle cell lymphoma after at least two prior lines of therapy, including a BTK inhibitor, under accelerated approval.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News