INDIANAPOLIS (dpa-AFX) - Eli Lilly and Company (LLY) announced positive topline results from the Phase 3 ACHIEVE-4 trial evaluating the efficacy and safety of Foundayo or orforglipron, compared to insulin glargine in adults with type 2 diabetes and obesity or overweight at increased cardiovascular risk. In the trial, Foundayo met the primary endpoint by demonstrating a non-inferior risk of major adverse cardiovascular events, including cardiovascular death, heart attack, stroke or hospitalization for unstable and sudden chest pain, compared to insulin glargine. Also, Foundayo showed superior improvements in A1C and body weight at 52 weeks vs. insulin glargine, which persisted through 104 weeks of therapy.
In the trial, the risk of cardiovascular death, heart attack, stroke, or hospitalization for unstable sudden chest pain was 16% lower for Foundayo vs. insulin glargine. The risk of all-cause death was 57% lower with Foundayo vs. insulin glargine.
Lilly will submit Foundayo for the treatment of type 2 diabetes to the FDA by the end of the second quarter under the Commissioner's National Priority Review Voucher.
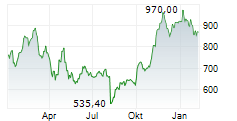
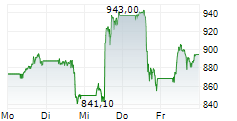
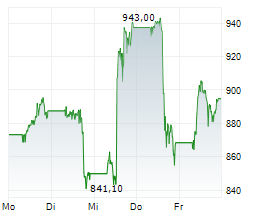
In pre-market trading on NYSE, Lilly shares are up 0.43 percent to $908.95.
For More Such Health News, visit rttnews.com.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News