KENILWORTH (NJ) (dpa-AFX) - Merck (MRK) announced the FDA granted priority review for two supplemental Biologics License Applications for KEYTRUDA and KEYTRUDA QLEX, Merck's anti-PD-1 therapy, each in combination with Padcev, for the treatment of patients with muscle-invasive bladder cancer who are eligible for cisplatin-based chemotherapy. The FDA set a PDUFA, or target action, date of August 17, 2026.
The sBLAs are based on data from the Phase 3 KEYNOTE-B15 trial, which was conducted in collaboration with Pfizer and Astellas. The companies plan to share the results with regulatory authorities worldwide for potential regulatory filings.
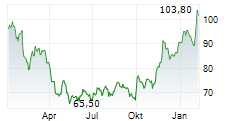
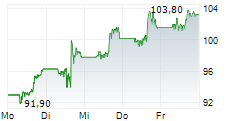
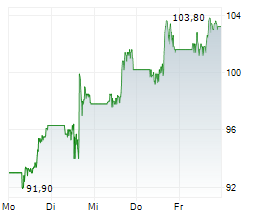
In pre-market trading on NYSE, Merck shares are down 0.46 percent to $118.52.
For More Such Health News, visit rttnews.com.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News