WASHINGTON (dpa-AFX) - AbbVie (ABBV) announced it received a Complete Response Letter from the FDA regarding the Biologics License Application for trenibotulinumtoxinE, a botulinum neurotoxin serotype E with a rapid onset of effect and short duration. The FDA requested additional information about manufacturing processes.
The company noted that the CRL does not identify any safety or efficacy concerns for TrenibotE, and does not request additional clinical studies. AbbVie expects to submit a thorough response in the coming months.
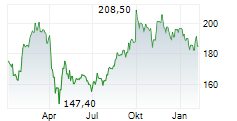
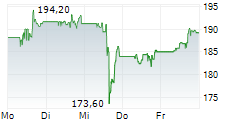
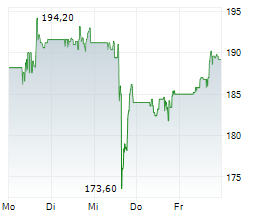
At last close, AbbVie shares were trading at $200.95, up 0.22%.
For More Such Health News, visit rttnews.com.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News