WASHINGTON (dpa-AFX) - AbbVie Inc. (ABBV) on Monday said it has submitted an application to the U.S. Food and Drug Administration seeking approval for a subcutaneous induction regimen of SKYRIZI for adults with moderately to severely active Crohn's disease.
The filing is backed by positive Phase 3 AFFIRM study results, which evaluated the efficacy and safety of subcutaneous SKYRIZI as an induction treatment, including in patients with prior advanced therapy failure.
SKYRIZI was first approved in 2022 for Crohn's disease, and AbbVie expects a decision on the new dosing regimen later this year.
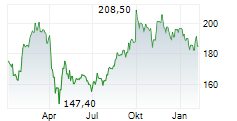
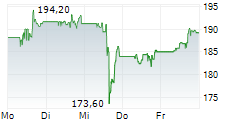
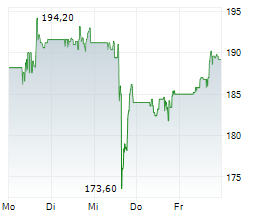
AbbVie shares closed at $198.71 on Friday, down 1.11%.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News