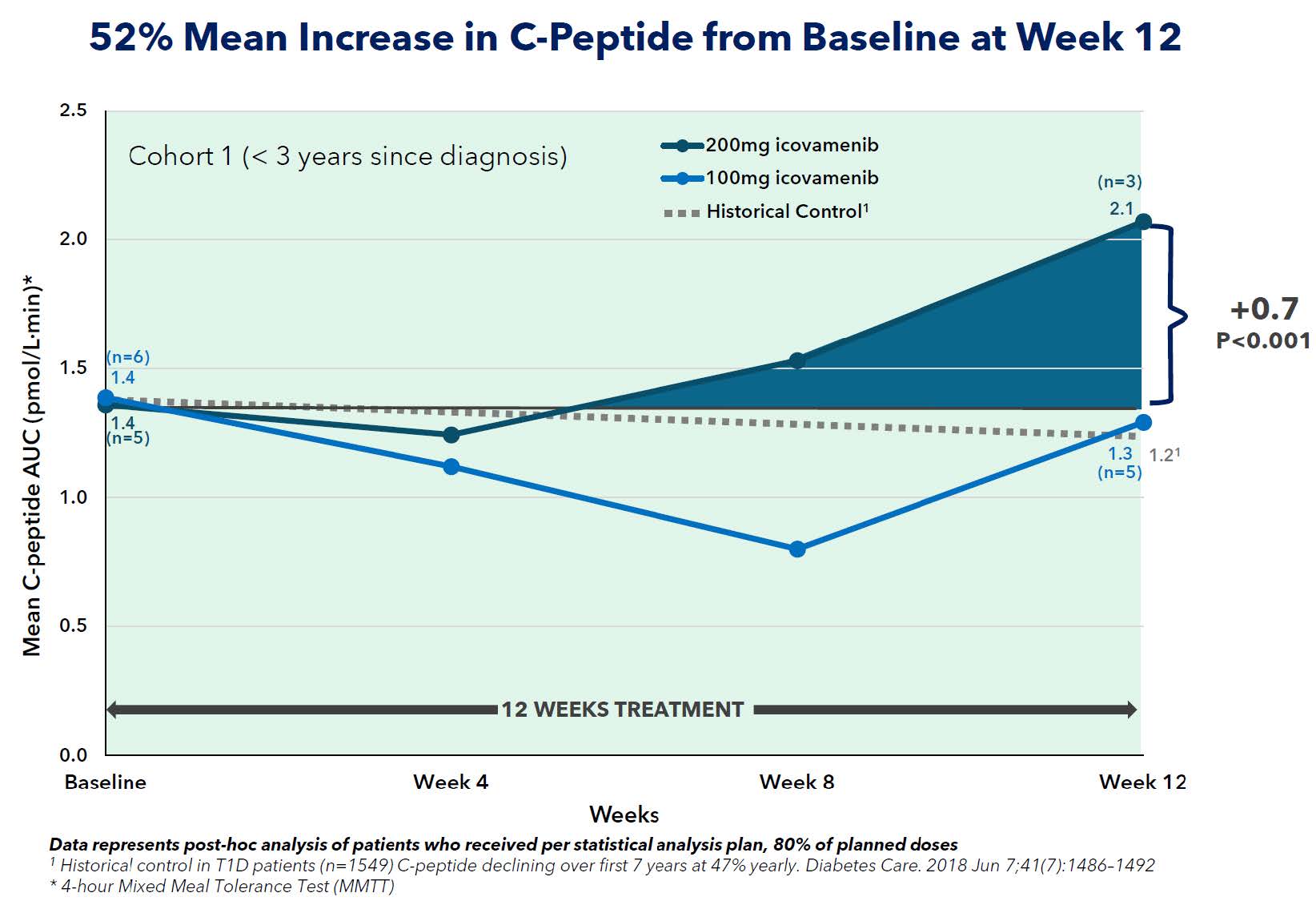
- A 52% increase from baseline in mean C-peptide AUC at Week 12 in patients diagnosed within 0-3 years (n=5) receiving icovamenib 200 mg, with a clear dose response observed vs 100 mg (n=6)
- Persistence observed through Week 52, with mean C-peptide AUC largely preserved in 200 mg group (~7% decline from baseline) following only 12 weeks of dosing
- Preservation of C-peptide also observed in patients diagnosed between 3-15 years (n=9)
- Icovamenib was generally well tolerated across all dosing arms and demonstrated a favorable safety and tolerability profile through Week 52
- Comprehensive dataset to be presented at the American Diabetes Association's (ADA) Scientific Sessions (abstract is preliminary until time of presentation; full release on June 5th at 6:30 pm CST)
- Company to host a conference call to discuss results on Tuesday, April 28 at 8:30 am ET
SAN CARLOS, Calif., April 27, 2026 (GLOBE NEWSWIRE) -- Biomea Fusion, Inc. ("Biomea" or "Biomea Fusion" or "the Company") (Nasdaq: BMEA), a clinical-stage diabetes and obesity company, today announced positive 52-week results from its Phase 2 COVALENT-112 trial evaluating the efficacy, safety, and tolerability of icovamenib in patients with type 1 diabetes ("T1D"). These data are based on a proof-of-concept study enrolling small subsets of Stage 3 T1D patients dosed with icovamenib at 100 mg and 200 mg in two cohorts (patients diagnosed within 3 years and those diagnosed within 3-15 years).
"The results we presented today mark an encouraging step forward for Biomea. The magnitude and durability observed are not typically seen in type 1 diabetes, which makes these findings particularly compelling. These data further validate targeting menin as a potential approach across both type 1 and type 2 diabetes," said Mick Hitchcock, Ph.D., Interim CEO and Board Member of Biomea Fusion. "We look forward to presenting additional data at an upcoming scientific meeting and advancing our type 1 diabetes program in collaboration with leading clinical centers in the United States"
The COVALENT-112 trial demonstrated encouraging results in patients with T1D. In patients diagnosed within 0-3 years, treatment with icovamenib 200 mg once daily for 12 weeks resulted in a 52% increase in mean C-peptide area under the curve (AUC) at Week 12 (p < 0.001; n=5), representing a magnitude of improvement that is not commonly reported in published studies of T1D. Importantly, the effect was durable following only 12 weeks of dosing, mean C-peptide AUC was largely preserved through Week 52, representing approximately a 7% decline from baseline. A dose response was observed, with the 200 mg dose demonstrating greater activity compared to 100 mg. Published natural history data suggest that patients with Stage 3 T1D typically experience substantial declines in C-peptide over time, underscoring the significance of preserved C-peptide following only a 12-week dosing period.
In patients with longer-standing disease (3-15 years since diagnosis), C-peptide levels were generally preserved through Week 52 (12-week treatment period + 40-week follow-up), with only a modest decline from baseline. A comprehensive dataset will be presented at the upcoming American Diabetes Association's (ADA) Scientific Sessions in June.
Icovamenib was generally well tolerated, with no new or unexpected safety signals identified throughout the 52-week observation period. Unlike investigational approaches in T1D that rely primarily on immune suppression or cellular transplantation, icovamenib is designed as a short course, orally administered therapy targeting beta cell biology, with effects that appear to persist beyond the treatment period.
Based on these data, Biomea, in collaboration with four U.S. academic centers, is planning a Phase 2 trial in patients with T1D diagnosed within the past 3 years. The study will evaluate whether extended dosing (up to 6 or 12 months) at 200 mg further improves C-peptide and whether the addition of an immunosuppressive agent enhances clinical outcomes. This study is planned to be initiated within the second half of this year at the Barbara Davis Center for Diabetes, Joslin Diabetes Center, UT Health San Antonio Diabetes Center, and the University of Miami Diabetes Research Institute.
"Efforts to intervene against T1D have historically focused on preserving remaining insulin secretion in people just diagnosed with T1D," said G. Alexander Fleming, MD, Founder & Executive Chairman of Kinexum and former FDA Senior Medical Officer and Division Leader for Metabolic & Endocrine Drugs, involved in the review of landmark diabetes and metabolic therapies including metformin, the first rapid acting insulin analogs, early statins, and PPAR agonists. "These icovamenib data are unique in showing increased C-peptide-reflected insulin secretion in patients with established T1D during dosing and persistence of this effect after treatment was stopped. In people with established T1D, endogenous insulin secretion progressively declines to very low levels. Any evidence of improvement in endogenous insulin secretion even among a few T1D individuals is unprecedented and of immense biologic and clinical significance. These findings warrant rigorous and longer-term evaluation."
COVALENT-112 Study Design
COVALENT-112 (NCT06152042) was an open label Phase 2 trial evaluating icovamenib in adult patients with T1D. The study enrolled patients aged 18 to 60 years with Stage 3 T1D, including those diagnosed within 0-3 years with residual beta cell function at baseline, defined by a screening C-peptide level =0.2 nmol/L (Cohort 1), as well as a broader population with disease duration of 3-15 years and residual beta cell function at baseline, defined by a screening C-peptide level =0.08 nmol/L (Cohort 2). Participants were assigned to receive icovamenib at 100 mg or 200 mg once daily for 12 weeks, followed by a 40-week post-treatment follow-up to assess durability of effect. Study enrollment and dosing were interrupted in May 2024 due to an FDA clinical hold, which was subsequently resolved. As a result, these data reflect approximately half of the originally intended patient population. A planned placebo-controlled Part 2 of the study was not completed.
The primary endpoint was the mean change from baseline in stimulated C-peptide area under the curve (AUC), measured during a mixed-meal tolerance test (MMTT), to evaluate endogenous insulin secretion. Secondary endpoints included additional measures of beta cell function, glycemic control, insulin use, and safety.
Conference Call and Webcast Details
Webcast of Biomea's investor update on Tuesday, April 28, 2026 at 8:30 am ET will be available to registered attendees under the Investors and Media section of the company's website at https://investors.biomeafusion.com/news-events/events. A replay of the presentation will be archived on Biomea's website following the event.
About Icovamenib
Icovamenib is an orally administered investigational small molecule currently in Phase 2 clinical development for the treatment of diabetes. Icovamenib targets menin, a transcriptional regulator implicated in beta cell dysfunction, and has been shown in preclinical and clinical studies to induce transient reductions in menin protein levels in pancreatic islets, thereby modulating pathways associated with insulin secretion and glycemic control. Through this mechanism, icovamenib has the potential to restore beta cell mass and function and improve endogenous insulin production. As a potential short-course therapy, icovamenib could represent a novel treatment approach for patients with diabetes, particularly those who have not achieved adequate control with standard-of-care therapies.
About Menin's Role in Diabetes
Loss of functional beta cell mass is a core component of the natural history in both major types of diabetes, T1D (mediated by autoimmune dysfunction) and T2D (mediated by metabolic dysfunction). Beta cells are found in the pancreas and are responsible for the synthesis and secretion of insulin. Insulin is a hormone that helps the body use glucose for energy and helps control blood glucose levels. In patients with diabetes, beta cell mass and function have been observed to be diminished, leading to insufficient insulin secretion and hyperglycemia. Menin is thought to act as a brake on beta cell turnover and growth, supporting the hypothesis that inhibition of menin may enable pathways associated with beta cell regeneration and improved function. Based on these and other scientific findings, Biomea is exploring the potential for icovamenib-mediated menin inhibition as a viable therapeutic approach to treat T1D and T2D.
About Type 1 Diabetes
Type 1 diabetes (T1D) is a chronic autoimmune disease in which the body's immune system destroys insulin-producing beta cells in the pancreas, leading to a loss of endogenous insulin production. Approximately 9.5 million people worldwide live with T1D, with an estimated 513,000 new diagnoses each year. In the United States, about 1.8 million individuals are affected. At diagnosis, patients often have already lost a significant portion of their functional beta cell mass, and this decline typically continues over time. As a result, individuals with T1D require lifelong insulin therapy and continuous glucose monitoring to manage blood sugar levels. Despite advances in care, T1D remains associated with meaningful risks, including severe hypoglycemia and diabetic ketoacidosis (DKA), as well as long-term complications such as cardiovascular disease, kidney disease, nerve damage, and vision loss. There are currently no approved therapies that address the underlying progressive loss of beta cell function in established (Stage 3) T1D beyond insulin replacement, highlighting a significant unmet medical need for disease-modifying treatments.
About Biomea Fusion
Biomea Fusion is a clinical-stage diabetes and obesity medicines company focused on the development of its oral small molecule therapies, icovamenib and BMF-650, for diabetes and obesity. These programs target metabolic disorders, a global health challenge affecting nearly half of all Americans and one-fifth of the world's population. Biomea's mission is to deliver transformative treatments that restore health for patients living with diabetes, obesity, and related conditions. We aim to cure!
Visit us at www.biomeafusion.com and follow us on LinkedIn X and Facebook
Forward-Looking Statements
Statements we make in this press release may include statements which are not historical facts and are considered forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended (the "Securities Act"), and Section 21E of the Securities Exchange Act of 1934, as amended (the "Exchange Act"). These statements may be identified by words such as "aims," "anticipates," "believes," "could," "estimates," "expects," "forecasts," "goal," "intends," "may," "plans," "possible," "potential," "seeks," "will," and variations of these words or similar expressions that are intended to identify forward-looking statements. Any such statements in this press release that are not statements of historical fact, including statements regarding the clinical and therapeutic potential of our product candidates and development programs, including icovamenib and the potential of icovamenib as a treatment for T1D and T2D, and our expectations regarding the optimal dose and target patient population; our research, development and regulatory plans; the mechanism of action of our product candidates and development programs; the progress and initiation of our ongoing and upcoming clinical trials, including our Phase 2COVALENT-112 trial; the anticipated availability of data from our clinical trials; our planned interactions with regulators, and the timing of such events may be deemed to be forward-looking statements. We intend these forward-looking statements to be covered by the safe harbor provisions for forward-looking statements contained in Section 27A of the Securities Act and Section 21E of the Exchange Act and are making this statement for purposes of complying with those safe harbor provisions. Any forward-looking statements in this press release are based on our current expectations, estimates and projections only as of the date of this release and are subject to a number of risks and uncertainties that could cause actual results to differ materially and adversely from those set forth in or implied by such forward-looking statements, including the risk that preliminary or interim results of preclinical studies or clinical trials may not be predictive of future or final results in connection with future clinical trials and the risk that we may encounter delays in preclinical or clinical development, patient enrollment and in the initiation, conduct and completion of our ongoing and planned clinical trials and other research and development activities. These risks concerning Biomea Fusion's business and operations are described in additional detail in its periodic filings with the U.S. Securities and Exchange Commission ("SEC"), including its most recent periodic report filed with the SEC and subsequent filings thereafter. Biomea Fusion explicitly disclaims any obligation to update any forward-looking statements except to the extent required by law.
Contact:
Meichiel Jennifer Weiss
Sr. Director of Investor Relations and Corporate Development
ir@biomeafusion.com
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/65d84e8f-dc21-4794-a041-80bf6b8ede2c