- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 11.05. | Biomea Fusion GAAP EPS of -$0.17 beats by $0.07 | 7 | Seeking Alpha | ||
| 11.05. | Biomea Fusion, Inc.: Biomea Fusion Reports First Quarter 2026 Financial Results and Corporate Highlights | 2.666 | GlobeNewswire (Europe) | Successfully completed chronic toxicology studies for icovamenib, providing the nonclinical support for advancing to chronic clinical dosing, beyond the 12-week clinical dosing that has been used... ► Artikel lesen | |
| 11.05. | Biomea Fusion, Inc. - 10-Q, Quarterly Report | - | SEC Filings | ||
| 11.05. | Biomea Fusion, Inc. - 8-K, Current Report | - | SEC Filings | ||
| 05.05. | Biomea Fusion, Inc.: Biomea Fusion Announces Poster Presentations of Icovamenib at the American Diabetes Association (ADA) 86th Scientific Sessions | 5 | GlobeNewswire (USA) | ||
| 05.05. | Citizens initiates Biomea Fusion stock coverage citing diabetes treatment potential | 2 | Investing.com | ||
| BIOMEA FUSION Aktie jetzt für 0€ handeln | |||||
| 28.04. | Biomea Fusion, Inc. - 8-K, Current Report | 1 | SEC Filings | ||
| 28.04. | Biomea reports positive type 1 diabetes trial results | 2 | Investing.com | ||
| 27.04. | Biomea: Positive Phase-2-Daten für Diabetes-Wirkstoff Icovamenib | 5 | Investing.com Deutsch | ||
| 27.04. | Biomea Fusion, Inc.: Biomea Fusion Announces Positive 52-Week Results from Phase 2 COVALENT-112 Trial in Type 1 Diabetes Showing C-Peptide Improvement and Durability Following 12-Weeks of Icovamenib Treatment | 324 | GlobeNewswire (Europe) | A 52% increase from baseline in mean C-peptide AUC at Week 12 in patients diagnosed within 0-3 years (n=5) receiving icovamenib 200 mg, with a clear dose response observed vs 100 mg (n=6)Persistence... ► Artikel lesen | |
| 31.03. | Biomea Fusion, Inc.: Biomea Fusion Announces First Patient Dosed in Newly Initiated Phase II Programs Enrolling Type 2 Diabetes Patients Failing on Standard-of-Care Therapies | 272 | GlobeNewswire (Europe) | COVALENT-211 Phase II enrolling insulin-deficient type 2 diabetes patients COVALENT-212 Phase II enrolling type 2 diabetes patients uncontrolled with a GLP-1 receptor agonist-based therapyTopline... ► Artikel lesen | |
| 25.03. | Analysts Sentiment on Biomea Fusion (BMEA) Remains Strong Amid Strong Diabetes Trial Results | 4 | Insider Monkey | ||
| 24.03. | Biomea Fusion GAAP EPS of -$1.18 beats by $0.36 | 5 | Seeking Alpha | ||
| 24.03. | Biomea Fusion, Inc. - 10-K, Annual Report | 1 | SEC Filings | ||
| 24.03. | Biomea Fusion, Inc.: Biomea Fusion Reports Full Year 2025 Financial Results and Corporate Highlights | 283 | GlobeNewswire (Europe) | Initiated two Phase II trials COVALENT-211 and COVALENT-212 (icovamenib in type 2 diabetes) with 26-week primary endpoint data anticipated in the fourth quarter of 2026Completed 52-week follow-up... ► Artikel lesen | |
| 24.03. | Biomea Fusion, Inc. - 8-K, Current Report | 1 | SEC Filings | ||
| 14.03. | Biomea Fusion Reports Positive Phase II Data For Diabetes Drug Icovamenib | 4 | Benzinga.com | ||
| 25.02. | Biomea Fusion, Inc. - 8-K, Current Report | 1 | SEC Filings | ||
| 14.01. | Biomea Fusion, Inc. - 8-K, Current Report | 2 | SEC Filings | ||
| 13.01. | Rodman & Renshaw initiates Biomea Fusion stock with Buy rating | 4 | Investing.com |
1 2 Weiter >>
28 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BIONTECH | 76,30 | -3,72 % | Vorsicht bei BioNTech! Novo Nordisk gibt Gas! Kaufchance bei Vidac Pharma Aktie! | Kaufstimmung bei Novo Nordisk. Die Aktie des dänischen Pharmagiganten scheint den seit Sommer 2024 begonnenen Abwärtstrend zu verlassen. Anleger reagieren auf positive Meldungen. Bei Vidac Pharma ist... ► Artikel lesen | |
| EVOTEC | 5,095 | -3,96 % | EQS-NVR: Evotec SE: Veröffentlichung der Gesamtzahl der Stimmrechte nach § 41 WpHG mit dem Ziel der europaweiten Verbreitung | EQS Gesamtstimmrechtsmitteilung: Evotec SE
/ Veröffentlichung der Gesamtzahl der Stimmrechte
Evotec SE: Veröffentlichung der Gesamtzahl der Stimmrechte nach § 41 WpHG mit dem... ► Artikel lesen | |
| BB BIOTECH | 46,150 | -2,02 % | BB Biotech: "Wachstumsstarke Mid- und Small Caps bilden das Rückgrat unseres Portfolios": Mehr Einzelwerte und ... | ||
| MEDIGENE | 0,027 | -6,21 % | PTA-Adhoc: Medigene AG: Widerruf der Zulassung der Aktien der Medigene AG zum Handel im regulierten Markt | DJ PTA-Adhoc: Medigene AG: Widerruf der Zulassung der Aktien der Medigene AG zum Handel im regulierten Markt
Veröffentlichung von Insiderinformationen gemäß Artikel 17 MAR Schlagwort(e): Sonstiges... ► Artikel lesen | |
| QIAGEN | 30,705 | -0,36 % | DEUTSCHE BANK RESEARCH stuft QIAGEN NV auf 'Buy' | FRANKFURT (dpa-AFX Analyser) - Deutsche Bank Research hat die Einstufung für Qiagen mit einem Kursziel von 43 US-Dollar auf "Buy" belassen. Das Interesse der Investoren habe vor allem dem Tuberkulosetest... ► Artikel lesen | |
| MODERNA | 38,365 | -3,13 % | Moderna, Inc.: Moderna to Present at Upcoming Conference in June 2026 | CAMBRIDGE, MA / ACCESS Newswire / June 2, 2026 / Moderna, Inc. (Nasdaq:MRNA), today announced its participation in the following upcoming investor conference:Goldman Sachs 47th Annual Global Healthcare... ► Artikel lesen | |
| PAION | 0,065 | -21,21 % | XFRA MISTRADE ANTRAG IN ISIN DE000A3E5EG5 WURDE STATTGEGEBEN | Dem Mistrade-Antrag wurde stattgegeben. Folgende Geschaefte in der ISIN DE000A3E5EG5, Wertpapier-Name: PAION AG INH O.N., wurden aufgehoben:ISIN Datum Zeit Volumen PreisDE000A3E5EG5 02.02.2026 08:13:40 300 0... ► Artikel lesen | |
| VALNEVA | 2,352 | -6,44 % | Original-Research: Valneva SE (von First Berlin Equity Research GmbH): Buy | Original-Research: Valneva SE - von First Berlin Equity Research GmbH
28.05.2026 / 17:08 CET/CEST
Veröffentlichung einer Research, übermittelt durch EQS News - ein Service der EQS Group.
Für den... ► Artikel lesen | |
| AMGEN | 280,90 | -0,72 % | Mesoblast: Während Amgen und Novo Nordisk den Biotech-Hype treiben, arbeitet dieser Zelltherapie-Player am operativen Durchbruch | ||
| EPIGENOMICS | 0,870 | 0,00 % | PTA-HV: Epigenomics AG: Einladung zur ordentlichen Hauptversammlung | DJ PTA-HV: Epigenomics AG: Einladung zur ordentlichen Hauptversammlung
Hauptversammlung gemäß -- 121 Abs. 4a AktG
Epigenomics AG: Einladung zur ordentlichen Hauptversammlung
Heidelberg... ► Artikel lesen | |
| NOVAVAX | 8,670 | -4,09 % | Novavax, Inc. (NVAX) Is a Trending Stock: Facts to Know Before Betting on It | ||
| STRYKER | 253,90 | -1,40 % | Stryker bringt das Pangea-Plattensystem auf den Markt und schließt den ersten Eingriff in Europa ab | Stryker (NYSE:SYK), ein weltweit führendes Unternehmen im Bereich Medizintechnik, gab heute die Markteinführung seines Pangea-Plattensystems in Europa bekannt, einer Plattensystem-Plattform zur Behandlung... ► Artikel lesen | |
| BIOGEN | 162,04 | -1,96 % | UCB and Biogen Announce Publication in The Lancet of Positive Dapirolizumab Pegol (DZP) Phase 3 Study Results in Systemic Lupus Erythematosus | Publication adds to previously reported findings from Phase 3 PHOENYCS GO study, demonstrating statistically significant improvement in disease activity at Week 48 with dapirolizumab pegol plus standard... ► Artikel lesen | |
| HEIDELBERG PHARMA | 2,680 | -2,19 % | EQS-HV: Heidelberg Pharma AG: Bekanntmachung der Einberufung zur Hauptversammlung am 23.06.2026 in Ladenburg mit dem Ziel der europaweiten Verbreitung gemäß §121 AktG | EQS-News: Heidelberg Pharma AG
/ Bekanntmachung der Einberufung zur Hauptversammlung
Heidelberg Pharma AG: Bekanntmachung der Einberufung zur Hauptversammlung am 23.06.2026 in... ► Artikel lesen | |
| ILLUMINA | 138,86 | -1,75 % | Guggenheim raises Illumina stock price target on clinical market strength |
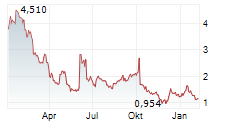
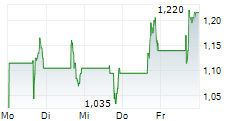
Das Unternehmen BIOMEA FUSION INC kann der Branche Biotechnologie zugeordnet werden. Die relative Kursveränderung beträgt aktuell 0,00 % bei einem Aktienkurs von 1,375 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu BIOMEA FUSION INC finden Sie auf Wallstreet Online.