NORTH CHICAGO (dpa-AFX) - Abbott Laboratories (ABT) on Tuesday said it received U.S. Food and Drug Administration (FDA) clearance and CE Mark for its next-generation Ultreon 3.0 AI-powered coronary imaging platform.
The system uses optical coherence tomography (OCT) to provide real-time guidance to doctors during minimally invasive procedures to open blocked heart arteries.
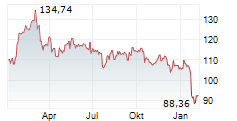
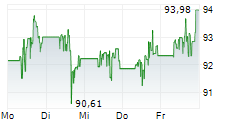
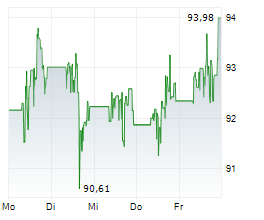
Abbott shares closed at $92.80 on Monday, up 1.83%.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News