INDIANAPOLIS (dpa-AFX) - Tuesday, Eli Lilly and Company (LLY) announced new long-term data from the LUCENT-3 open-label extension study in patients with moderately to severely active ulcerative colitis treated with Omvoh.
The study showed that 63.5 percent of Omvoh-treated patients who achieved disease clearance at one year sustained it at four years.
Moreover, the long-term safety profile in patients with moderately to severely active UC was consistent with the known safety profile of Omvoh with no new safety signals observed.
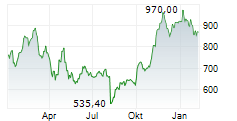
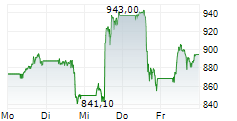
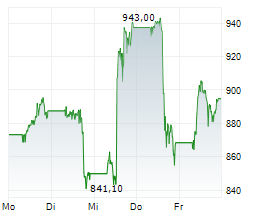
In the pre-market hours, LLY is trading at $966.06, down 0.23 percent on the New York Stock Exchange.
Copyright(c) 2026 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2026 AFX News