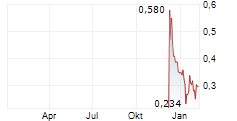
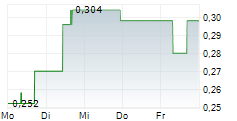
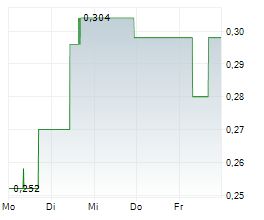
Vancouver, British Columbia--(Newsfile Corp. - May 12, 2026) - DiagnosTear Technologies Inc. (CSE: DTR) (FSE: X8F) ("DiagnosTear" or the "Company"), a leader in rapid, eye-based diagnostic technologies, is pleased to announce that on May 12, 2026, it has entered into a definitive exclusive global license agreement for the commercialization of the CLARIFY eye imaging platform, A breakthrough technology enabling blood analysis through a simple eye scan, eliminating the need for needles and traditional blood draws.
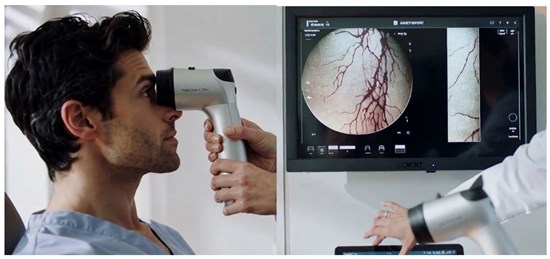
Illustrative rendering of CLARIFY's eye-based imaging technology (design subject to final development)
To view an enhanced version of this graphic, please visit:
https://images.newsfilecorp.com/files/11754/297199_diagnostear.jpg
CLARIFY integrates advanced optical imaging, spectral analysis, and AI-driven algorithms to read signals from tiny blood vessels in the eye and translate them into useful health information. If successfully developed and clinically validated, CLARIFY could provide instant measurement of critical blood markers such as red blood cells, hemoglobin and potentially white blood cells and other biomarkers through a quick, needle-free eye scan. Additionally, CLARIFY offers platform-level optionality and can support multiple diagnostic applications over time, with potential use across primary care, emergency settings, chronic disease monitoring, neonatology settings, and remote or at-home healthcare. This creates a pathway toward a scalable, repeat-use diagnostic solution with the potential to establish recurring, high-margin software-driven revenue streams. The underlying technology has already demonstrated early feasibility, measuring red blood cells, hemoglobin and other blood parameters non-invasively using a handheld prototype device.
Under the agreement, signed with Sheba Impact (Sheba Medical Center) and Ramot (Tel Aviv University), DiagnosTear receives exclusive worldwide rights to develop, manufacture, and commercialize CLARIFY-based products, while the licensors retain ownership of the underlying intellectual property.
Key Terms:
- 3% royalty on annual net sales up to US$20M; 4% thereafter
- 15% sublicensing revenue share pre-approval; 12% post-regulatory approval and with respect to royalties the greater of these or half of the royalties mentioned above
DiagnosTear will lead development, including clinical validation, regulatory strategy, and commercialization, with the objective of advancing a scalable, AI-driven diagnostic platform toward market.
"This agreement secures exclusive access to a technology that could redefine how blood diagnostics are performed at some selected common clinical scenarios," said Dr. Shimon Gross, CEO of DiagnosTear. "A future where a physician can obtain information on the very basic blood composition by rapid, non-invasive imaging of the eye surface has enormous clinical and commercial potential. Partnering with Sheba Medical Center with its clinical capabilities and Tel Aviv University with its deep knowledge in AI and physics allows us to accelerate development of this next generation diagnostic platform."
"We believe the eye represents an untapped gateway to diagnostics of systemic clinical conditions," said Mr. Yaacov Michlin, Chairman of DiagnosTear. "CLARIFY brings together cutting-edge optics, large vascular datasets, and AI-driven analysis to create an entirely new diagnostic category. This is also part of our strategy to expand DiagnosTear footprint and products portfolio in the points of care."
About DiagnosTear Technologies Inc.
DiagnosTear is a pioneering medical diagnostics company developing rapid, point-of-care diagnostic technologies using the eye as a source of biological information. By leveraging tear analysis and advanced ocular imaging, the Company is developing solutions to improve the detection and differentiation of Dry Eye, Red Eye, and systemic conditions detectable through ocular biomarkers. DiagnosTear's technologies aim to make diagnostics faster, more accessible, and less invasive.
For media inquiries, please contact:
Shimon Gross
DiagnosTear Technologies Inc.
Email: info@diagnostear.com
Website: www.diagnostear.com
Phone number: +972-52-3408550
THE CANADIAN SECURITIES EXCHANGE HAS NOT REVIEWED AND DOES NOT ACCEPT RESPONSIBILITY FOR THE ACCURACY OR ADEQUACY OF THIS RELEASE, NOR HAS OR DOES THE CSE'S REGULATION SERVICES PROVIDER.
Forward-Looking Statements
Forward-looking statements in this news release include, but are not limited to, statements regarding: the anticipated benefits of the license agreement with Sheba Impact and Ramot; the development, clinical validation, regulatory approval, manufacturing, and commercialization of the CLARIFY eye imaging platform and related products; the ability of CLARIFY to enable needle-free blood analysis and measure blood markers such as red and white blood cells and hemoglobin; the potential clinical applications of CLARIFY across primary care, emergency settings, chronic disease monitoring, and remote or at-home healthcare; the potential to generate recurring, high-margin software-driven revenue streams; the scalability of the platform; the Company's regulatory strategy and timing of commercialization; the timing and amount of royalty and other payments under the license agreement; and the Company's business plans, strategy, and objectives generally.
Forward-looking statements are based on a number of assumptions and estimates that, while considered reasonable by management of the Company as of the date of this news release, are inherently subject to significant business, economic, competitive, regulatory, scientific, and technological uncertainties and contingencies. Key assumptions include, among others: that the CLARIFY technology will perform as expected in further development and clinical testing; that the Company will be able to successfully develop, validate, manufacture, and commercialize CLARIFY-based products; that required regulatory approvals will be obtained on a timely basis or at all; that the Company will have access to sufficient capital to fund its development and commercialization activities; that the Company will maintain good standing under its license agreement; and that market demand, competitive conditions, and general economic conditions will remain consistent with current expectations.
Forward-looking statements involve known and unknown risks, uncertainties, and other factors that may cause actual results, performance, or achievements to differ materially from those expressed or implied by such statements. Such factors include, but are not limited to: risks relating to the early stage of the Company's technology and products; the outcome and timing of preclinical studies, clinical trials, and regulatory reviews; the risk that CLARIFY may not achieve the accuracy, reliability, or clinical utility required for regulatory approval or market acceptance; risks related to intellectual property owned by third parties and licensed to the Company, including the Company's continued ability to maintain its license rights; dependence on third-party licensors, collaborators, and suppliers; competition from other diagnostic technologies and companies; the need for additional financing and the Company's ability to obtain such financing on acceptable terms; risks related to the protection and enforcement of intellectual property rights; changes in laws, regulations, and healthcare reimbursement policies; general market, political, and economic conditions; and the other risks and uncertainties identified in the Company's public disclosure documents filed on SEDAR+ at www.sedarplus.ca.
Although the Company has attempted to identify important factors that could cause actual results to differ materially from those contained in forward-looking statements, there may be other factors that cause results not to be as anticipated, estimated, or intended. There can be no assurance that such statements will prove to be accurate, as actual results and future events could differ materially from those anticipated in such statements. Readers are cautioned not to place undue reliance on forward-looking statements.
Forward-looking statements are made as of the date of this news release, and the Company disclaims any intention or obligation to update or revise any forward-looking statements, whether as a result of new information, future events, or otherwise, except as required by applicable law.

To view the source version of this press release, please visit https://www.newsfilecorp.com/release/297199
Source: DiagnosTear Technologies Inc.