| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
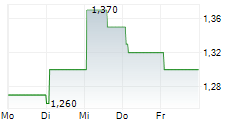
| 1,210 | 1,240 | 13:04 |
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 28.04. | ABBISKO-B (02256): NEXT DAY DISCLOSURE RETURNS | - | HKEx | ||
| 28.04. | ABBISKO-B (02256): COMPLETION OF PLACING OF NEW SHARES UNDER THE GENERAL MANDATE | - | HKEx | ||
| 28.04. | ABBISKO-B (02256): NOTIFICATION LETTER AND REPLY FORM TO NON-REGISTERED SHAREHOLDERS - REMINDER LETTER REGARDING THE ARRANGEMENT OF ELECTRONIC DISSEMINATION ... | - | HKEx | ||
| ABBISKO CAYMAN Aktie jetzt für 0€ handeln | |||||
| 28.04. | ABBISKO-B (02256): NOTIFICATION LETTER AND REPLY FORM TO REGISTERED SHAREHOLDERS - REMINDER LETTER REGARDING THE ARRANGEMENT OF ELECTRONIC DISSEMINATION ... | - | HKEx | ||
| 28.04. | ABBISKO-B (02256): FORM OF PROXY FOR THE 2025 ANNUAL GENERAL MEETING | - | HKEx | ||
| 28.04. | ABBISKO-B (02256): NOTICE OF THE ANNUAL GENERAL MEETING | - | HKEx | ||
| 28.04. | ABBISKO-B (02256): PROPOSALS FOR GRANTING OF GENERAL MANDATES TO ISSUE SHARES AND TO REPURCHASE SHARES RE-ELECTION OF DIRECTORS RE-APPOINTMENT OF AUDITOR ... | 1 | HKEx | ||
| 28.04. | ABBISKO-B (02256): PROPOSED AMENDMENTS TO THE TENTH AMENDED AND RESTATED MEMORANDUM AND ARTICLES OF ASSOCIATION AND THE ADOPTION OF THE ELEVENTH AMENDED ... | - | HKEx | ||
| 28.04. | ABBISKO-B (02256): 2025 ENVIRONMENTAL, SOCIAL AND GOVERNANCE REPORT | - | HKEx | ||
| 28.04. | ABBISKO-B (02256): 2025 ANNUAL REPORT | - | HKEx | ||
| 23.04. | ABBISKO-B Subsidiary Presents 6 Preclinical and Translational Research Findings at AACR | 2 | AASTOCKS | ||
| 23.04. | ABBISKO-B (02256): VOLUNTARY ANNOUNCEMENT - ABBISKO THERAPEUTICS SHOWCASES SIX RESEARCH ADVANCES AT 2026 AACR, HIGHLIGHTING PAN-KRAS, 4TH GENERATION EGFR, ... | 1 | HKEx | ||
| 21.04. | ABBISKO-B (02256): PLACING OF NEW SHARES UNDER THE GENERAL MANDATE | 2 | HKEx | ||
| 08.04. | ABBISKO-B (02256): GRANT OF OPTIONS UNDER THE POST-IPO SHARE OPTION SCHEME AND GRANT OF RSUS UNDER THE 2019 SHARE INCENTIVE PLAN | 1 | HKEx | ||
| 31.03. | ABBISKO-B (02256): VOLUNTARY ANNOUNCEMENT - FGFR4 INHIBITOR IRPAGRATINIB GRANTED ORPHAN DRUG DESIGNATION BY EMA FOR HEPATOCELLULAR CARCINOMA | 2 | HKEx | ||
| 31.03. | Abbisko Therapeutics Co., Ltd.: Abbisko Therapeutics' FGFR2/3 Inhibitor ABSK061 Receives FDA IND Clearance for Achondroplasia in Children | 108 | PR Newswire | SHANGHAI, March 31, 2026 /PRNewswire/ -- On 30 March, 2026, Abbisko Therapeutics Co., Ltd. ("Abbisko Therapeutics" hereafter, HKEX code: 02256.HK) announced that... ► Artikel lesen | |
| 30.03. | ABBISKO-B (02256): VOLUNTARY ANNOUNCEMENT - FGFR2/3 INHIBITOR ABSK061 RECEIVES FDA IND CLEARANCE FOR ACHONDROPLASIA IN CHILDREN | 1 | HKEx | ||
| 19.03. | ABBISKO-B (02256): VOLUNTARY ANNOUNCEMENT - FGFR2/3 INHIBITOR ABSK061 GRANTED ORPHAN DRUG DESIGNATION BY FDA FOR ACHONDROPLASIA | 6 | HKEx | ||
| 16.03. | ABBISKO-B (02256): VOLUNTARY ANNOUNCEMENT - ABBISKO THERAPEUTICS' IRPAGRATINIB COMBINED WITH TARGETED-IMMUNOTHERAPY COMPLETES FIRST PATIENT DOSING IN ... | 1 | HKEx | ||
| 11.03. | ABBISKO-B (02256): VOLUNTARY ANNOUNCEMENT - FDA GRANTED RARE PEDIATRIC DISEASE DESIGNATION FOR FGFR2/3 INHIBITOR ABSK061 | 8 | HKEx |
1 2 Weiter >>
38 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| QIAGEN | 28,465 | -0,44 % | Qiagen erhöht Dividende und stellt weitere Aktienrückkäufe in Aussicht | DJ Qiagen erhöht Dividende und stellt weitere Aktienrückkäufe in Aussicht
DOW JONES--Die Aktionäre von Qiagen können sich über eine höhere Dividende freuen. Wie es in der Einladung zur Hauptversammlung... ► Artikel lesen | |
| TANGO THERAPEUTICS | 20,560 | 0,00 % | Tango Therapeutics, Inc.: Tango Therapeutics Reports First Quarter 2026 Financial Results and Provides Business Highlights | First clinical data from PRMT5/RAS(ON) combination trial to be presented in 2026 Cash position of $380 million as of March 31, 2026, with runway into 2028 beyond anticipated key data inflection points... ► Artikel lesen | |
| HCW BIOLOGICS | 1,210 | -0,82 % | HCW Biologics Inc.: HCW Biologics Reports First Quarter 2026 Business Highlights and Financial Results | MIRAMAR, Fla., May 14, 2026 (GLOBE NEWSWIRE) -- HCW Biologics Inc. (the "Company" or "HCW Biologics") (NASDAQ: HCWB), a clinical-stage biopharmaceutical company focused on developing transformative... ► Artikel lesen | |
| APOGEE THERAPEUTICS | 81,09 | -0,06 % | Jefferies raises Apogee Therapeutics stock price target on trial outlook | ||
| PRAXIS PRECISION MEDICINES | 341,89 | -0,20 % | Praxis Precision Medicines, Inc. - 8-K, Current Report | ||
| CG ONCOLOGY | 66,93 | 0,00 % | CG Oncology Inc.: CG Oncology Reports Positive First Results from CORE-008 Cohort CX Phase 2 Trial Evaluating Intravesical Combination Therapy in High-Risk BCG-Exposed and BCG-Unresponsive Patients | - High CR rates at any time observed in the CIS-containing population with 85.7% and 92.3% in the ITT population and Efficacy Evaluable population, respectively - High-Grade - EFS in the overall intention-to-treat... ► Artikel lesen | |
| EVOTEC | 4,618 | -1,74 % | Evotec zapft den Kapitalmarkt an | Evotec platziert Wandelanleihen über 116,1 Mio. EUR und finanziert damit das Transformationsprogramm "Project Horizon". Die Evotec SE hat sich frisches Kapital über den Wandelanleihemarkt gesichert.... ► Artikel lesen | |
| BIONTECH | 76,40 | -2,98 % | Community Rückblick: wO-User zu BioNTech Quartalsbericht: "Oh wow, die Zahlen sind eine Katastrophe!" | © Foto: picture alliance / Laci Perenyi | Laci PerenyiBioNTech steht nach neuen Quartalszahlen und angekündigten Standortschließungen weiter unter Druck. Hohe Verluste, Umbau des Geschäfts und das Aus... ► Artikel lesen | |
| COGENT BIOSCIENCES | 32,710 | +0,09 % | Cogent Biosciences, Inc.: Cogent Biosciences Announces Multiple Presentations at the European Hematology Association (EHA) 2026 Congress | ||
| NUVALENT | 102,40 | +0,10 % | Nuvalent, Inc.: Nuvalent Highlights Recent Pipeline Progress, Reiterates Key Anticipated Milestones, and Reports First Quarter 2026 Financial Results | NDA submitted for neladalkib in TKI pre-treated advanced ALK-positive NSCLC
NDA for zidesamtinib in TKI pre-treated advanced ROS1-positive NSCLC under FDA review... ► Artikel lesen | |
| JADE BIOSCIENCES | 23,355 | 0,00 % | Jade Biosciences Reports First Quarter 2026 Financial Results and Provides Corporate Update | Interim biomarker-rich Phase 1 data for JADE101 to be featured in an oral session at the 63rd European Renal Association Congress Phase 2 trial of JADE101 in IgA nephropathy patients expected to begin... ► Artikel lesen | |
| RELAY THERAPEUTICS | 12,205 | +0,04 % | H.C. Wainwright is Bullish on Relay Therapeutics, Inc. (RLAY) | ||
| TYRA BIOSCIENCES | 37,070 | +0,08 % | Tyra Biosciences GAAP EPS of -$0.64 | ||
| KYMERA THERAPEUTICS | 82,25 | +0,05 % | Kymera presents KT-621 atopic dermatitis trial data at conferences | ||
| BEAM THERAPEUTICS | 27,945 | +0,05 % | H.C. Wainwright reiterates Beam Therapeutics stock rating at buy |
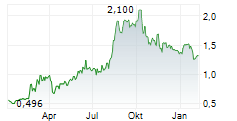
Das Unternehmen ABBISKO CAYMAN LTD kann der Branche Biotechnologie zugeordnet werden. Mit einer aktuellen Kursveränderung von 0,000 (0,00 %) liegt der Kurs bei 1,170 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu ABBISKO CAYMAN LTD finden Sie auf Wallstreet Online.