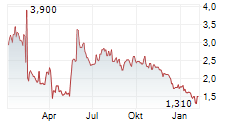
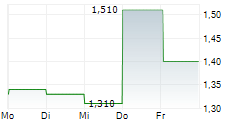
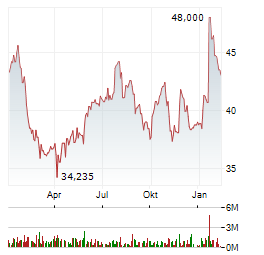
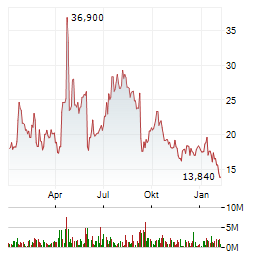
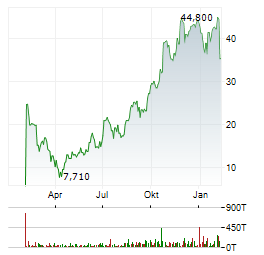
Chart-Typ:
Zeitraum:
| QIAGEN N.V.: QIAGEN Supports Infectious Disease Research and Surveillance Efforts for Bundibugyo Ebolavirus Outbreak | Two custom-designed research-use-only (RUO) QIAcuity digital PCR assays now available to target Bundibugyo ebolavirus sequences associated with the current outbreak Assays designed for... ► Artikel lesen |
| QIAGEN N.V.: QIAGEN unterstützt Forschungs- und Überwachungsmaßnahmen zum Ausbruch des Bundibugyo-Ebolavirus | Ab sofort stehen zwei maßgeschneiderte QIAcuity digitale PCR-Assays für den Einsatz in der Forschung (Research Use Only, RUO) zur Verfügung, die auf die mit dem aktuellen Ausbruch in Verbindung... ► Artikel lesen |
| DEUTSCHE BANK RESEARCH stuft QIAGEN NV auf 'Buy' | FRANKFURT (dpa-AFX Analyser) - Deutsche Bank Research hat die Einstufung für Qiagen mit einem Kursziel von 43 US-Dollar auf "Buy" belassen. Das Interesse der Investoren habe vor allem dem Tuberkulosetest... ► Artikel lesen |
| Sionna Therapeutics, Inc.: Sionna Therapeutics Completes Enrollment in PreciSION CF Phase 2a Trial Evaluating NBD1 Stabilizer, SION-719, Added to Standard of Care in Participants with Cystic Fibrosis | WALTHAM, Mass., April 27, 2026 (GLOBE NEWSWIRE) -- Sionna Therapeutics, Inc. (Nasdaq: SION), a clinical-stage biopharmaceutical company on a mission to revolutionize the current treatment paradigm... ► Artikel lesen |
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu ANNEXIN PHARMACEUTICALS AB finden Sie auf Wallstreet Online.