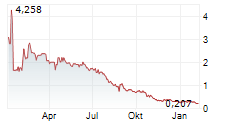
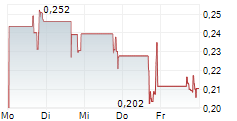
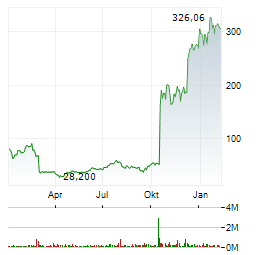
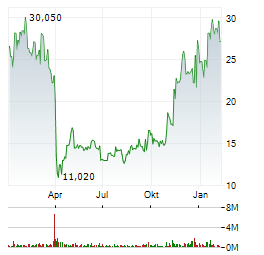
Chart-Typ:
Zeitraum:
| Praxis Precision Medicines, Inc.: Praxis Precision Medicines Provides Vormatrigine Program Update | POWER1 Study in highly refractory patients with focal onset seizures did not meet its primary success measure Secondary measure, the 50% response rate, was met and seizure reduction during the second... ► Artikel lesen |
| Praxis Precision Medicines, Inc.: Praxis Precision Medicines Provides Corporate Update and Reports First Quarter 2026 Financial Results | FDA accepted the new drug application (NDA) for ulixacaltamide in Essential Tremor with a PDUFA target action date of January 29, 2027, and the NDA for relutrigine, with priority review, in SCN2A... ► Artikel lesen |
| Praxis Precision Medicines, Inc.: Praxis Precision Medicines Announces FDA Acceptance of New Drug Application for Ulixacaltamide HCl in Patients with Essential Tremor | FDA assigned PDUFA target action date of January 29, 2027 No advisory committee meeting expected BOSTON, April 14, 2026 (GLOBE NEWSWIRE) -- Praxis Precision Medicines, Inc. (NASDAQ: PRAX), a fully... ► Artikel lesen |
| Servier to Acquire Edgewise Therapeutics' Muscular Dystrophy Business | Acquisition supports Servier's strategic ambition in rare neurology for patients with high unmet medical needs.Transaction includes sevasemten, a late-stage investigational treatment for... ► Artikel lesen |
| Edgewise Therapeutics Reports First Quarter 2026 Financial Results and Advances Clinical Programs in Muscular Dystrophy and Cardiovascular Indications | - Announced positive long-term sevasemten data demonstrating sustained functional stabilization in Becker muscular dystrophy (Becker) up to 3.5 years of follow-up... ► Artikel lesen |
| Edgewise Therapeutics Reports Fourth Quarter and Full Year 2025 Financial Results with Strong Progress Across Muscular Dystrophy and Cardiovascular Programs | - CIRRUS-HCM 12-week data of EDG-7500 in obstructive and nonobstructive hypertrophic cardiomyopathy (HCM) expected in H1 2026 -
- Phase 1 healthy adult trial... ► Artikel lesen |
| Jade Biosciences Announces Positive Interim Phase 1 Results for JADE101 Demonstrating Potential Best-in-Class Profile and Every-12-Week Dosing in IgA Nephropathy | ~70% IgA reductions sustained at 12 weeks after a single dose of JADE101>70% IgA reductions projected at steady state with a single subcutaneous maintenance injection every 12 weeksIgA-lowering effect... ► Artikel lesen |
| Jade Biosciences Announces First Participant Dosed in First-in-Human Phase 1 Clinical Trial of JADE201, a Novel Half-Life Extended Afucosylated Anti-BAFF Receptor Monoclonal Antibody | Interim Phase 1 data in rheumatoid arthritis expected in 2027 are anticipated to inform indication prioritization across multiple potential autoimmune indications supported by BAFF-R biology JADE201... ► Artikel lesen |
| Jade Biosciences Announces First Participant Dosed in Phase 2 JUNIPER Clinical Trial of JADE101 in IgA Nephropathy | Interim Phase 2 JADE101 data are expected in 2027 SAN FRANCISCO and VANCOUVER, British Columbia, May 26, 2026 (GLOBE NEWSWIRE) -- Jade Biosciences, Inc. (the "Company" or "Jade") (Nasdaq: JBIO), a... ► Artikel lesen |
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu AZITRA INC finden Sie auf Wallstreet Online.