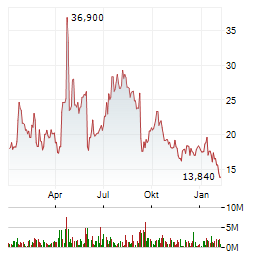
Chart-Typ:
Zeitraum:
| Tango Therapeutics, Inc.: Tango Therapeutics Reports First Quarter 2026 Financial Results and Provides Business Highlights | First clinical data from PRMT5/RAS(ON) combination trial to be presented in 2026 Cash position of $380 million as of March 31, 2026, with runway into 2028 beyond anticipated key data inflection points... ► Artikel lesen |
| Tango Therapeutics Sub, Inc: Tango Therapeutics Announces Leadership Appointments to Accelerate Late-Stage Development of Vopimetostat | - Matthew Gall appointed Chief Financial Officer -- Additional key appointments add expertise in clinical operations, corporate strategy and project leadership - BOSTON, April 15, 2026 (GLOBE... ► Artikel lesen |
| Erasca, Inc.: Erasca and Tango Therapeutics Enter into Clinical Collaboration to Evaluate Combination of ERAS-0015 and Vopimetostat | ERAS-0015, a pan-RAS molecular glue, will be evaluated in combination with PRMT5 inhibitor vopimetostat Tango will sponsor the clinical trial and Erasca will supply ERAS-0015 at no cost SAN DIEGO... ► Artikel lesen |
| Biotech-Partnerschaft: Roche greift zu: Dieser Milliarden-Deal lässt Anleger aufhorchen | © Foto: Vladimir Borovic - Adobe StockRoche steigt mit einem milliardenschweren Deal bei Nurix ein. Im Fokus steht ein vielversprechender Wirkstoff gegen Krebs, Autoimmun- und Nervenkrankheiten.Der... ► Artikel lesen |
| Roche And Nurix Ink $2.3 Bln Collaboration To Co-Develop BTK Degrader Bexobrutideg | SOUTH SAN FRANCISCO (dpa-AFX) - Roche (RHHBY) announced it has entered into an exclusive licensing and collaboration agreement with Nurix Therapeutics, Inc. (NRIX). The two companies will co-develop... ► Artikel lesen |
| Roche geht 2,3-Milliarden-Dollar-Deal mit Nurix für Blutkrebsmittel ein | DJ Roche geht 2,3-Milliarden-Dollar-Deal mit Nurix für Blutkrebsmittel ein
Von Nina Kienle
DOW JONES--Roche hat mit dem US-Biotechunternehmen Nurix Therapeutics eine exklusive Lizenz- und Kooperationsvereinbarung... ► Artikel lesen |
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu ME THERAPEUTICS HOLDINGS INC finden Sie auf Wallstreet Online.