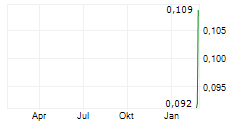
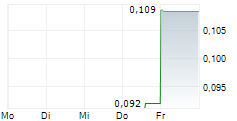
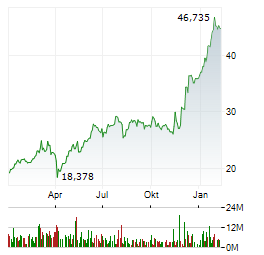
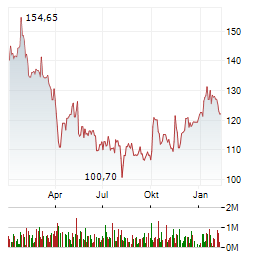
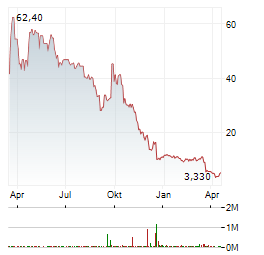
Chart-Typ:
Zeitraum:
| Dax vor turbulentem Wochenausklang: Bayer - Das wird jetzt für die Aktie entscheidend | Dem Dax steht einmal mehr ein turbulenter Wochenausklang bevor. Bislang gestaltete sich die Handelswoche positiv. Ein robuster Wochenausklang würde das zuletzt zu beobachtende Erholungsszenario bestätigen... ► Artikel lesen |
| Bayer-Medikament Kerendia in China gegen Herzinsuffizienz zugelassen | DJ Bayer-Medikament Kerendia in China gegen Herzinsuffizienz zugelassen
DOW JONES--DOW JONES--Bayer hat für sein Medikament Kerendia in China die Zulassung für eine weitere Indikation bekommen.... ► Artikel lesen |
| DAX-Check LIVE: Bayer, DHL Group, Lanxess, Rheinmetall, SMA Solar im Fokus | Der deutsche Aktienmarkt hat am Donnerstag seine jüngste Gewinnserie beendet. Der DAX, der sich am Vormittag noch leicht nach oben vorgetastet hatte, drehte mangels Anschlusskäufen ins Minus und schloss... ► Artikel lesen |
| EQS-PVR: Merck KGaA: Release according to Article 40, Section 1 of the WpHG [the German Securities Trading Act] with the objective of Europe-wide distribution | EQS Voting Rights Announcement: Merck KGaA
Merck KGaA: Release according to Article 40, Section 1 of the WpHG [the German Securities Trading Act] with the objective of Europe-wide... ► Artikel lesen |
| MilliporeSigma Launches Supplier Sustainability Toolkits to Help Advance Supply Chain Impact | NORTHAMPTON, MA / ACCESS Newswire / May 21, 2026 / MilliporeSigma, the U.S. and Canada Life Science business of Merck KGaA, Darmstadt, Germany, has launched its Sustainability Toolkits, making four... ► Artikel lesen |
| EQS-PVR: Merck KGaA: Release according to Article 40, Section 1 of the WpHG [the German Securities Trading Act] with the objective of Europe-wide distribution | EQS Voting Rights Announcement: Merck KGaA
Merck KGaA: Release according to Article 40, Section 1 of the WpHG [the German Securities Trading Act] with the objective of Europe-wide... ► Artikel lesen |
| Akari Therapeutics Plc: Akari Therapeutics Reports Breakthrough Preclinical Data Demonstrating Synergistic Activity of AKTX-101 with KRAS Inhibition in KRAS-Mutated Pancreatic Cancer Models | An ADC with a novel RNA spliceosome modulating payload PH1 paired with a KRAS inhibitor demonstrates synergistic cytotoxic activity against KRAS G12D and G12C- driven pancreatic cancer modelsData... ► Artikel lesen |
| Akari Therapeutics Plc: Akari Therapeutics Secures Key European Patent For Its Novel RNA Splicing Modulator ADC Payload, Strengthening Global IP Estate For All Major Global Markets | TAMPA, Fla. and LONDON, May 12, 2026 (GLOBE NEWSWIRE) -- Akari Therapeutics, Plc (Nasdaq: AKTX), an oncology biotechnology company developing antibody drug conjugates (ADCs) with novel RNA splicing... ► Artikel lesen |
| Akari Therapeutics Plc: Akari Therapeutics Secures Australian Patent Approval, Further Expanding Global Protection of Its Proprietary PH1 RNA Splicing Modulator ADC Payload | Patent covers proprietary 2nd generation Thailanstatin analog payload designed for use in ADCs (antibody-drug conjugates) targeting cancerApproval strengthens Akari's global intellectual property... ► Artikel lesen |
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu NEUROTHERA LABS INC finden Sie auf Wallstreet Online.