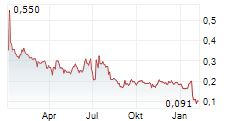
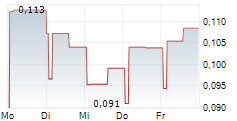
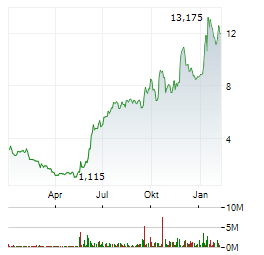
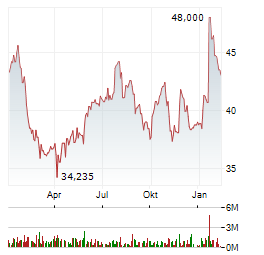
Chart-Typ:
Zeitraum:
| Parabilis Medicines, Inc.: Parabilis Medicines Announces Pricing of Upsized Initial Public Offering | CAMBRIDGE, Mass., June 09, 2026 (GLOBE NEWSWIRE) -- Parabilis Medicines, Inc. (Nasdaq: PBLS) ("Parabilis"), a clinical-stage biopharmaceutical company built to develop transformative medicines addressing... ► Artikel lesen |
| Regeneron Pharmaceuticals, Inc.: Regeneron Announces Strategic Collaboration with Parabilis Medicines to Advance Novel Antibody-Helicon Conjugates Across Multiple Therapeutic Areas | The multi-target collaboration combines Regeneron's industry-leading antibody capabilities with Parabilis' novel Helicon peptide platform Agreement provides for Parabilis to receive $125M from Regeneron... ► Artikel lesen |
| Starke Studiendaten: Nach 626% Kursplus in einem Jahr: Dieser Krebsspezialist macht Anleger sprachlos | © Foto: Bernd Weißbrod/dpa Ein Wirkstoff gegen die tödlichste Form von Bauchspeicheldrüsenkrebs zeigt Daten, die selbst erfahrene Onkologen aufhorchen lassen. Die Aktie reagierte entsprechend.Pankreas-Karzinom... ► Artikel lesen |
| Tango Therapeutics, Inc.: Tango Therapeutics Reports First Quarter 2026 Financial Results and Provides Business Highlights | First clinical data from PRMT5/RAS(ON) combination trial to be presented in 2026 Cash position of $380 million as of March 31, 2026, with runway into 2028 beyond anticipated key data inflection points... ► Artikel lesen |
| Tango Therapeutics Sub, Inc: Tango Therapeutics Announces Leadership Appointments to Accelerate Late-Stage Development of Vopimetostat | - Matthew Gall appointed Chief Financial Officer -- Additional key appointments add expertise in clinical operations, corporate strategy and project leadership - BOSTON, April 15, 2026 (GLOBE... ► Artikel lesen |
| DEUTSCHE BANK RESEARCH stuft QIAGEN NV auf 'Buy' | FRANKFURT (dpa-AFX Analyser) - Deutsche Bank Research hat Qiagen mit einem Kursziel von 43 US-Dollar auf "Buy" belassen. Das Feedback aus Gesprächen mit etlichen US-Laborexperten deute darauf hin, dass... ► Artikel lesen |
| QIAGEN N.V.: QIAGEN unterstützt Forschungs- und Überwachungsmaßnahmen zum Ausbruch des Bundibugyo-Ebolavirus | Ab sofort stehen zwei maßgeschneiderte QIAcuity digitale PCR-Assays für den Einsatz in der Forschung (Research Use Only, RUO) zur Verfügung, die auf die mit dem aktuellen Ausbruch in Verbindung... ► Artikel lesen |
| QIAGEN N.V.: QIAGEN Supports Infectious Disease Research and Surveillance Efforts for Bundibugyo Ebolavirus Outbreak | Two custom-designed research-use-only (RUO) QIAcuity digital PCR assays now available to target Bundibugyo ebolavirus sequences associated with the current outbreak Assays designed for... ► Artikel lesen |
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu PILA PHARMA AB finden Sie auf Wallstreet Online.