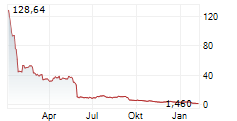
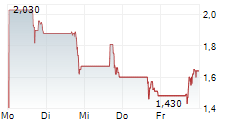
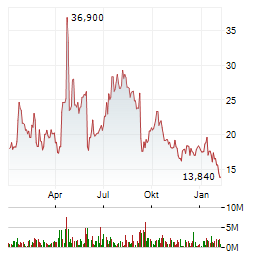
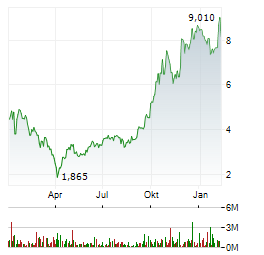
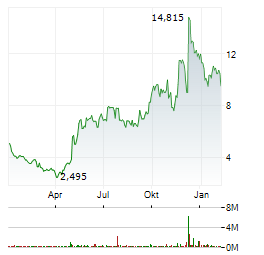
Chart-Typ:
Zeitraum:
| Relay Therapeutics, Inc.: Relay Therapeutics Announces Pricing of Public Offering of Common Stock | CAMBRIDGE, Mass., May 20, 2026 (GLOBE NEWSWIRE) -- Relay Therapeutics, Inc. (Nasdaq: RLAY), a clinical-stage, small molecule precision medicine company developing potentially life-changing therapies... ► Artikel lesen |
| Relay Therapeutics, Inc.: Relay Therapeutics Announces Initial Clinical Data Demonstrating That Zovegalisib Has Potential for Differentiated Safety and Efficacy in Patients with PIK3CA-Driven Vascular Anomalies | Promising initial efficacy data with 60% volumetric response rate across doses and 29%* at the lowest tested dose of 100mg twice daily (BID) with all patients ongoing Interim investigator- and patient-reported... ► Artikel lesen |
| Relay Therapeutics, Inc.: Relay Therapeutics Reports First Quarter 2026 Financial Results and Corporate Updates | FDA Breakthrough Therapy designation granted to zovegalisib for PIK3CA-mutant, HR+/HER2- advanced breast cancer, the Phase 3 ReDiscover-2 trial population in 2L breast cancer Presented zovegalisib... ► Artikel lesen |
| Fulcrum Therapeutics, Inc.: Fulcrum Therapeutics Announces Discontinuation of Pociredir Program in Sickle Cell Disease and Initiation of Strategic Review | ? Decision follows FDA feedback regarding the implications of the secondary malignancies observed with Tazverik- (tazemetostat) and the product's subsequent global withdrawal on the benefit-risk... ► Artikel lesen |
| Fulcrum Therapeutics, Inc.: Fulcrum Therapeutics Announces Recent Business Highlights and Financial Results for First Quarter 2026 | - Presented positive clinical data for pociredir, demonstrating robust and rapid fetal hemoglobin (HbF) induction, improvements in markers of hemolysis and anemia, and encouraging trends in vaso-occlusive... ► Artikel lesen |
| Fulcrum Therapeutics, Inc.: Fulcrum Therapeutics Announces Positive 12-Week Results from the 20 mg Dose Cohort of the Phase 1b PIONEER Trial of Pociredir in Sickle Cell Disease | ? Mean absolute fetal hemoglobin (HbF) increased by 12.2% (from 7.1% to 19.3%) at Week 12 (n=12), representing a rapid, robust, and clinically relevant response, with progression toward pan-cellular... ► Artikel lesen |
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu REVELATION BIOSCIENCES INC finden Sie auf Wallstreet Online.