NEW YORK CITY, NY / ACCESS Newswire / July 9, 2025 / Pomerantz LLP is investigating claims on behalf of investors of Sanofi ("Sanofi" or the "Company") (NASDAQ:SNY). Such investors are advised to contact Danielle Peyton at newaction@pomlaw.com or 646-581-9980, ext. 7980.
The investigation concerns whether Sanofi and certain of its officers and/or directors have engaged in securities fraud or other unlawful business practices.
[Click here for information about joining the class action]
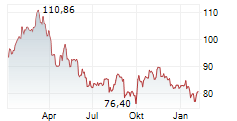
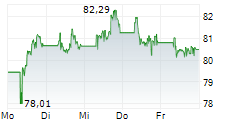
On May 30, 2025, Sanofi issued a press release announced mixed data from a Phase 3 program for itepekimab, an antibody therapy targeting the lung disorder chronic obstructive pulmonary disease. Although the AERIFY-1 late-stage trial met the primary endpoint with a statistically significant and clinically meaningful impact, the AERIFY-2 trial failed to reach the same goal.
On this news, Sanofi's American Depositary Receipt ("ADR") price fell $2.98 per ADR, or 5.69%, to close at $49.37 per ADR on May 30, 2025.
The Pomerantz Firm, with offices in New York, Chicago, Los Angeles, London, and Paris is acknowledged as one of the premier firms in the areas of corporate, securities, and antitrust class litigation. Founded by the late Abraham L. Pomerantz, known as the dean of the class action bar, the Pomerantz Firm pioneered the field of securities class actions. Today, more than 80 years later, the Pomerantz Firm continues in the tradition he established, fighting for the rights of the victims of securities fraud, breaches of fiduciary duty, and corporate misconduct. The Firm has recovered numerous multimillion-dollar damages awards on behalf of class members. See www.pomerantzlaw.com.
Attorney advertising. Prior results do not guarantee similar outcomes.
SOURCE: Pomerantz LLP
View the original press release on ACCESS Newswire:
https://www.accessnewswire.com/newsroom/en/business-and-professional-services/investor-alert-pomerantz-law-firm-investigates-claims-on-behalf-1046910