| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
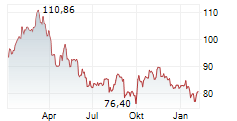
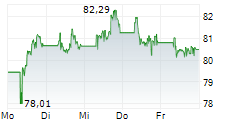
| 72,87 | 72,93 | 19:23 | |
| 72,85 | 72,95 | 19:23 |
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| Di | Bicara Therapeutics hires Replimune, Sanofi alum as chief commercial officer | 24 | FiercePharma | ||
| Di | Sanofi-Aktie: Jetzt günstig genug? | 626 | sharedeals.de | Die Aktie von Sanofi geriet in den letzten Monaten deutlich unter Druck. Gleichzeitig mehren sich jedoch die Anzeichen, dass der Pharmakonzern operativ vor einer deutlich stärkeren Wachstumsphase steht.... ► Artikel lesen | |
| So | Mesoblast Aktie: Zelltherapie im Aufwind - warum der kleine Biotech-Player plötzlich in einer Liga mit Merck, Regeneron & Co. gedacht werden muss | Small- & Micro Cap Investment | |||
| 06.05. | Sanofi asks to pull Tzield bid from FDA's controversial CNPV program: report | 56 | FiercePharma | ||
| 05.05. | Sanofi expands AI capabilities, investing $294M to scale Toronto hub | 28 | FiercePharma | ||
| 05.05. | Dividendenbekanntmachungen (05.05.2026) | 7.841 | wallstreetONLINE (Dividenden) | Unternehmen ISIN-Code Dividende (Währung) Dividende (EUR) 1ST SOURCE CORPORATION US3369011032 0,43 USD 0,3678 EUR AGRICULTURAL BANK OF CHINA LTD ADR US00850M1027 0,4702 USD 0,4022 EUR ALCON... ► Artikel lesen | |
| SANOFI Aktie jetzt für 0€ handeln | |||||
| 05.05. | Sanofi - 6-K, Report of foreign issuer | 7 | SEC Filings | ||
| 05.05. | JPMORGAN stuft SANOFI auf 'Neutral' | 1.036 | dpa-AFX-Analyser | NEW YORK (dpa-AFX Analyser) - Die US-Bank JPMorgan hat die Einstufung für Sanofi nach dem Quartalsbericht des Pharmakonzerns auf "Neutral" mit einem Kursziel von 95 Euro belassen. Die Ziele für 2026... ► Artikel lesen | |
| 04.05. | Biopharma giant Sanofi to grow Toronto presence with expanded AI centre | 43 | BetaKit | ||
| 04.05. | Sanofi invests $294M to expand AI Center of Excellence in Toronto and accelerate innovation for patients | 18 | CNW | ||
| 01.05. | Merck KgaA's New CEO Kai Beckmann Takes Charge As Belén Garijo Becomes Sanofi CEO | 779 | AFX News | PARIS (dpa-AFX) - German science and technology major Merck KGaA's (MKGAY, MRK.DE) new Chief Executive Officer Kai Beckmann took charge on Friday, succeeding Belén Garijo, who joined French... ► Artikel lesen | |
| 30.04. | Belén Garijo wird Direktorin und CEO von Sanofi | 54 | Bionity.com | ||
| 29.04. | Sanofi: Annual General Meeting of April 29, 2026 - Belén Garijo appointed as Director and Chief Executive Officer of Sanofi | 803 | GlobeNewswire (Europe) | Annual General Meeting of April 29, 2026
Belén Garijo appointed as Director and Chief Executive Officer of Sanofi
Paris, April 29, 2026. Sanofi's Mixed General Meeting of Shareholders convened... ► Artikel lesen | |
| 29.04. | Sanofi's Tzield gets expanded FDA approval for early-stage type 1 diabetes | 10 | PMLiVE | ||
| 28.04. | Giants Costco, Sanofi, and SAP Raise Dividends by Over 10% | 70 | MarketBeat | ||
| 28.04. | Von Merck zu Sanofi: Lieber Schlangengrube als Ruhestand, Frau Garijo? | 115 | FAZ | ||
| 28.04. | BERNSTEIN RESEARCH stuft SANOFI auf 'Outperform' | 1.005 | dpa-AFX-Analyser | NEW YORK (dpa-AFX Analyser) - Das US-Analysehaus Bernstein Research hat das Kursziel für Sanofi von 110 auf 107 Euro gesenkt, aber die Einstufung auf "Outperform" belassen. Die neue Chefin habe zwar... ► Artikel lesen | |
| 28.04. | DEUTSCHE BANK RESEARCH stuft SANOFI auf 'Buy' | 814 | dpa-AFX-Analyser | FRANKFURT (dpa-AFX Analyser) - Deutsche Bank Research hat Sanofi mit einem Kursziel von 100 Euro auf "Buy" belassen. Emmanuel Papadakis passte seine Schätzungen für den Pharmakonzern an die ordentlichen... ► Artikel lesen | |
| 27.04. | UBS stuft SANOFI auf 'Neutral' | 1.056 | dpa-AFX-Analyser | ZÜRICH (dpa-AFX Analyser) - Die Schweizer Großbank UBS hat die Einstufung für Sanofi mit einem Kursziel von 88 Euro auf "Neutral" belassen. Analyst Matthew Weston hatte schon mit einer starken Entwicklung... ► Artikel lesen | |
| 24.04. | Sanofi: Sanofi successfully prices 2.3 billion of bond issue | 1.047 | GlobeNewswire (Europe) | Sanofi successfully prices 2.3 billion of bond issue
Paris, April 24, 2026 - Sanofi announces that it has successfully priced its offering of €2.3 billion of notes across 3 tranches:
€1,000... ► Artikel lesen |
1 2 3 4 5 Weiter >>
787 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BAYER | 37,970 | -1,22 % | Bayer zahlt bis zu 2,45 Milliarden Dollar für Übernahme im Bereich Augenheilkunde | Der DAX-notierte Bayer-Konzern übernimmt Perfuse Therapeutics vollständig und sichert sich damit die weltweiten Rechte an PER-001, einem niedermolekularen Endothelinrezeptor-Antagonisten in der klinischen... ► Artikel lesen | |
| NOVO NORDISK | 40,025 | -0,07 % | Novo Nordisk mit Lichtblick: Nimmt die Trendwende jetzt endlich Fahrt auf? | Die Aktie des dänischen Pharmakonzerns Novo Nordisk arbeitet seit geraumer Zeit an der Ausbildung eines tragfähigen Bodens respektive an einer unteren Trendwende. Die letzten Monate haben der Aktie... ► Artikel lesen | |
| PFIZER | 22,025 | -0,11 % | JEFFERIES stuft PFIZER INC auf 'Buy' | NEW YORK (dpa-AFX Analyser) - Das Analysehaus Jefferies hat Pfizer mit einem Kursziel von 35 US-Dollar auf "Buy" belassen. Der Pharmakonzern habe im ersten Quartal positiv überrascht und die Jahresziele... ► Artikel lesen | |
| NOVARTIS | 127,28 | +0,97 % | Biotech und die Effizienz-Revolution: Warum für Novartis, BioNTech und BioNxt die letzte Meile entscheidet | Während für Biotech-Investoren über Jahrzehnte hinweg das Dogma galt, dass nur die Entdeckung eines völlig neuen Moleküls den Weg zu einem Milliarden-Exit ebnen kann, zeigt die Realität des Marktes... ► Artikel lesen | |
| MERCK KGAA | 121,15 | +7,21 % | Merck trotz Gegenwind: Aktie schießt nach Prognose-Anhebung in die Höhe | Die Aktie des Darmstädter Merck-Konzerns legt nach einer überraschenden Prognoseanhebung deutlich zu. Trotz widriger Wechselkurse und rückläufiger Medikamentenumsätze überzeugt das Unternehmen mit robusten... ► Artikel lesen | |
| GILEAD SCIENCES | 114,02 | -0,82 % | Gilead: Erwartungen übertroffen - darum fällt die Aktie trotzdem | Die Biotech-Gesellschaft Gilead Sciences hat am Donnerstagabend nach US-Börsenschluss die Zahlen für das erste Quartal des neuen Geschäftsjahres veröffentlicht. Trotz besser als erwarteter Vorgaben... ► Artikel lesen | |
| AURORA CANNABIS | 2,908 | +1,47 % | AURORA CANNABIS INC: Ruhe vor dem nächsten Impuls | ||
| GSK | 21,720 | -0,32 % | GSK findet für neues Hepatitis-Medikament Vertriebspartner in China | DJ GSK findet für neues Hepatitis-Medikament Vertriebspartner in China
Von Adria Calatayud
DOW JONES--GSK kooperiert mit Sino Biopharmaceutical, um die Markteinführung für sein Hepatitis-B-Medikament... ► Artikel lesen | |
| ABBVIE | 177,30 | +0,11 % | Ihre wichtigsten Termine: Heute im Fokus: AbbVie, Colgate-Palmolive, Toyota, Sony, Krones, Bechtle, Evonik | © Foto: Hiro Komae/AP/dpaGut geplant in den Tag. Mit dem wO-Tagesausblick haben Sie die wichtigsten Termine im Blick und starten bestens vorbereitet in den neuen Handelstag.Unternehmenstermine 05:00... ► Artikel lesen | |
| ROCHE | 348,85 | -0,13 % | Roche greift nach KI-Schatz: Milliarden-Deal: Roche setzt in Diagnostik auf KI | © Foto: Georgios Kefalas/KEYSTONE/dpaRoche zahlt 1,05 Milliarden US-Dollar für PathAI, um Diagnostik mit KI zu verbessern.Roche gab bekannt, dass sie sich mit PathAI auf einen Kaufpreis von 1,05 Milliarden... ► Artikel lesen | |
| CANOPY GROWTH | 0,950 | +1,95 % | CANOPY GROWTH CORPORATION: Struktur im Stillstand | ||
| STADA ARZNEIMITTEL | - | - | Enzymatica AB: Quarterly report Q1/2026 - Enzymatica strengthens international position through STADA agreement | First quarter
Net sales amounted to SEK 11.4 million (12.3).
Operating result amounted to SEK -13.6.million (-18.0)
Earnings per share, before... ► Artikel lesen | |
| ELI LILLY | 866,30 | +2,73 % | Eli Lilly, Compass Pathways und das Nadelöhr der Psychiatrie: Emyria Limited lanciert globale Infrastruktur-Lösung | ||
| MERCK & CO | 97,19 | +1,54 % | Mesoblast Aktie: Warum sich der Blick auf diesen Biotech-Player jetzt lohnt - und was Anleger von Merck, Regeneron & Co. lernen können | ||
| ASTRAZENECA | 158,45 | +0,80 % | AstraZeneca und OMP demonstrieren auf dem Gartner Supply Chain Symposium/Xpo 2026 Planung im Tempo des Wandels | Wie eine schnelle Entscheidungsfindung die Lücke zwischen Erkenntnis und Handeln in komplexen, schnelllebigen Lieferketten
schließt
ANTWERPEN, BE / ACCESS Newswire / 7. Mai 2026... ► Artikel lesen |
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu SANOFI SA finden Sie auf Wallstreet Online.