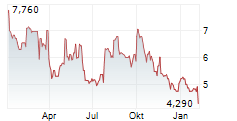
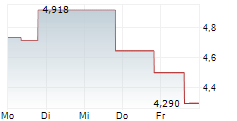
OSE Immunotherapeutics has made tangible progress across its clinical development pipeline, creating positive momentum, in our view. In immuno-inflammation, monoclonal antibody therapy lusvertikimab showed positive results in Phase II for ulcerative colitis (UC), and in immuno-oncology, cancer vaccine Tedopi is progressing through the registrational Phase III ARTEMIA trial in non-small cell lung cancer (NSCLC). While these are the two leading proprietary candidates, we highlight that OSE also has a strong track record of establishing fruitful partnerships with key pharma players with other pipeline candidates, adding to the company's value proposition. Overall, OSE's activities span multiple disease areas, providing a 'portfolio of a pipeline', which we aim to provide an overview of in this note. Readers may refer to our April outlook note for a discussion on financials and valuation.Den vollständigen Artikel lesen ...
© 2025 Edison Investment Research