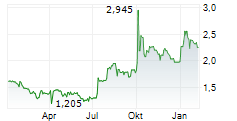
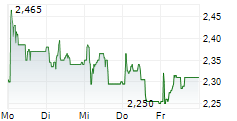
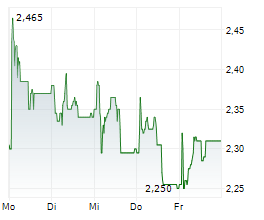
Herantis Pharma has made tangible progress towards raising funds to support its Phase II proof-of-concept study for lead asset HER-096 as a potential disease-modifier for Parkinson's disease (PD). In early February, the company successfully completed a directed issue amounting to €4.2m (gross proceeds). This funding will be used to support ongoing discussions with prospective pharma partners and make the required preparations, including regulatory clearance, to commence this subsequent clinical trial. More recently, it announced that it had been selected for an €8.0m grant (subject to final negotiations) from the Horizon Europe 2025 Research and Innovation programme, adding non-dilutive funding to support the Phase II study. In our view, these two sources of funding provide external validation of Herantis's approach to addressing a significant unmet medical need in the field of PD.Den vollständigen Artikel lesen ...
© 2026 Edison Investment Research