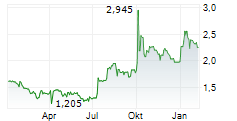
Herantis Pharma's FY25 results reflect positive momentum for HER-096, which is being developed as a potential disease-modifier for Parkinson's disease (PD). Notably, the candidate successfully completed the Phase Ib trial, meeting primary and secondary endpoints. The associated biomarker programme also added to a robust foundation for Phase II, in our view. With a year-end gross cash position of €2.6m, plus a directed share issue of €4.2m (gross) in February 2026, Herantis has a cash runway into Q127 (according to management guidance). While this is not sufficient to execute the Phase II trial, it should cover the remaining preparations. Management is actively seeking partners to continue development activities, but is also exploring both equity and grant financing options.Den vollständigen Artikel lesen ...
© 2026 Edison Investment Research